N-methyl-D-aspartate antibody encephalitis: temporal progression of clinical and paraclinical observations in a predominantly non-paraneoplastic disorder of both sexes
- PMID: 20511282
- PMCID: PMC2877907
- DOI: 10.1093/brain/awq113
N-methyl-D-aspartate antibody encephalitis: temporal progression of clinical and paraclinical observations in a predominantly non-paraneoplastic disorder of both sexes
Abstract
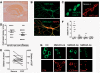
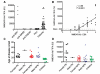
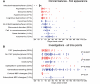
Antibodies to the N-methyl-d-aspartate subtype of glutamate receptor have been associated with a newly-described encephalopathy that has been mainly identified in young females with ovarian tumours. However, the full clinical spectrum and treatment responses are not yet clear. We established a sensitive cell-based assay for detection of N-methyl-d-aspartate receptor antibodies in serum or cerebrospinal fluid, and a quantitative fluorescent immunoprecipitation assay for serial studies. Although there was marked intrathecal synthesis of N-methyl-d-aspartate receptor antibodies, the absolute levels of N-methyl-d-aspartate receptor antibodies were higher in serum than in cerebrospinal fluid. N-methyl-d-aspartate receptor antibodies were of the immunoglobulin G1 subclass and were able to activate complement on N-methyl d-aspartate receptor-expressing human embryonic kidney cells. From questionnaires returned on 44 N-methyl-d-aspartate receptor antibody-positive patients, we identified a high proportion without a detected tumour (35/44, 80%: follow-up 3.6-121 months, median 16 months). Among the latter were 15 adult females (43%), 10 adult males (29%) and 10 children (29%), with four in the first decade of life. Overall, there was a high proportion (29%) of non-Caucasians. Good clinical outcomes, as defined by reductions in modified Rankin scores, correlated with decreased N-methyl-d-aspartate receptor antibody levels and were associated with early (<40 days) administration of immunotherapies in non-paraneoplastic patients (P < 0.0001) and earlier tumour removal in paraneoplastic patients (P = 0.02). Ten patients (23%) who were first diagnosed during relapses had no evidence of tumours but had received minimal or no immunotherapy during earlier episodes. Temporal analysis of the onset of the neurological features suggested progression through two main stages. The time of onset of the early features, characterized by neuropsychiatric symptoms and seizures preceded by a median of 10-20 days, the onset of movement disorders, reduction in consciousness and dysautonomia. This temporal dichotomy was also seen in the timing of cerebrospinal fluid, electroencephalographic and in the rather infrequent cerebral imaging changes. Overall, our data support a model in which the early features are associated with cerebrospinal fluid lymphocytosis, and the later features with appearance of oligoclonal bands. The immunological events and neuronal mechanisms underlying these observations need to be explored further, but one possibility is that the early stage represents diffusion of serum antibodies into the cortical grey matter, whereas the later stage results from secondary expansion of the immunological repertoire within the intrathecal compartment acting on subcortical neurons. Four patients, who only had temporal lobe epilepsy without oligoclonal bands, may represent restriction to the first stage.
Figures



References
-
- Bien CG, Urbach H, Schramm J, Soeder BM, Becker AJ, Voltz R, et al. Limbic encephalitis as a precipitating event in adult-onset temporal lobe epilepsy. Neurology. 2007;69:1236–44. - PubMed
-
- Buckley C, Oger J, Clover L, Tuzun E, Carpenter K, Jackson M, et al. Potassium channel antibodies in two patients with reversible limbic encephalitis. Ann Neurol. 2001;50:73–8. - PubMed
-
- Castillo P, Woodruff B, Caselli R, Vernino S, Lucchinetti C, Swanson J, et al. Steroid-responsive encephalopathy associated with autoimmune thyroiditis. Arch Neurol. 2006;63:197–202. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

