An autoimmune-mediated strategy for prophylactic breast cancer vaccination
- PMID: 20512124
- PMCID: PMC3095829
- DOI: 10.1038/nm.2161
An autoimmune-mediated strategy for prophylactic breast cancer vaccination
Abstract
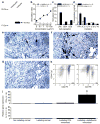
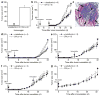
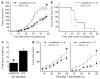
Although vaccination is most effective when used to prevent disease, cancer vaccine development has focused predominantly on providing therapy against established growing tumors. The difficulty in developing prophylactic cancer vaccines is primarily due to the fact that tumor antigens are variations of self proteins and would probably mediate profound autoimmune complications if used in a preventive vaccine setting. Here we use several mouse breast cancer models to define a prototypic strategy for prophylactic cancer vaccination. We selected alpha-lactalbumin as our target vaccine autoantigen because it is a breast-specific differentiation protein expressed in high amounts in the majority of human breast carcinomas and in mammary epithelial cells only during lactation. We found that immunoreactivity against alpha-lactalbumin provides substantial protection and therapy against growth of autochthonous tumors in transgenic mouse models of breast cancer and against 4T1 transplantable breast tumors in BALB/c mice. Because alpha-lactalbumin is conditionally expressed only during lactation, vaccination-induced prophylaxis occurs without any detectable inflammation in normal nonlactating breast tissue. Thus, alpha-lactalbumin vaccination may provide safe and effective protection against the development of breast cancer for women in their post-child-bearing, premenopausal years, when lactation is readily avoidable and risk for developing breast cancer is high.
Conflict of interest statement
The authors have no conflicting or competing financial interests regarding publication of this manuscript.
Figures

Comment in
-
Cancer: steps towards a prophylactic breast cancer vaccine.Nat Rev Drug Discov. 2010 Aug;9(8):594. doi: 10.1038/nrd3233. Nat Rev Drug Discov. 2010. PMID: 20671760 No abstract available.
Similar articles
-
Experimental autoimmune breast failure: a model for lactation insufficiency, postnatal nutritional deprivation, and prophylactic breast cancer vaccination.Am J Pathol. 2012 Sep;181(3):775-84. doi: 10.1016/j.ajpath.2012.05.025. Am J Pathol. 2012. PMID: 22901749 Free PMC article.
-
A prophylactic vaccine for breast cancer?Breast Cancer Res. 2010;12(4):310. doi: 10.1186/bcr2628. Epub 2010 Aug 31. Breast Cancer Res. 2010. PMID: 20828424 Free PMC article.
-
A prophylactic vaccine for breast cancer? Why not?Breast Cancer Res. 2010;12(6):405. doi: 10.1186/bcr2775. Epub 2010 Dec 8. Breast Cancer Res. 2010. PMID: 21143772 Free PMC article. No abstract available.
-
Challenges to the development of antigen-specific breast cancer vaccines.Breast Cancer Res. 2001;3(2):95-8. doi: 10.1186/bcr278. Epub 2001 Jan 11. Breast Cancer Res. 2001. PMID: 11250753 Free PMC article. Review.
-
Primary immunoprevention of adult onset cancers by vaccinating against retired tissue-specific self-proteins.Semin Immunol. 2020 Feb;47:101392. doi: 10.1016/j.smim.2020.101392. Epub 2020 Jan 8. Semin Immunol. 2020. PMID: 31926646 Review.
Cited by
-
Management of breast cancer by vaccine: fact or fiction.J IMA. 2013 Apr 22;44(1):44-1-9055. doi: 10.5915/44-1-9055. Print 2012. J IMA. 2013. PMID: 23864997 Free PMC article.
-
Advances in immunotherapy for triple-negative breast cancer.Mol Cancer. 2023 Sep 2;22(1):145. doi: 10.1186/s12943-023-01850-7. Mol Cancer. 2023. PMID: 37660039 Free PMC article. Review.
-
Combination of sunitinib with anti-tumor vaccination inhibits T cell priming and requires careful scheduling to achieve productive immunotherapy.Int J Cancer. 2014 Apr 1;134(7):1695-705. doi: 10.1002/ijc.28488. Epub 2013 Oct 8. Int J Cancer. 2014. PMID: 24105638 Free PMC article.
-
Advanced Oxidation Nanoprocessing Boosts Immunogenicity of Whole Tumor Cells.Adv Sci (Weinh). 2023 Aug;10(22):e2302250. doi: 10.1002/advs.202302250. Epub 2023 May 21. Adv Sci (Weinh). 2023. PMID: 37211712 Free PMC article.
-
Cancer Vaccines for Triple-Negative Breast Cancer: A Systematic Review.Vaccines (Basel). 2023 Jan 9;11(1):146. doi: 10.3390/vaccines11010146. Vaccines (Basel). 2023. PMID: 36679991 Free PMC article. Review.
References
-
- Lollini PL, Cavallo F, Nanni P, Forni G. Vaccines for tumour prevention. Nat Rev Cancer. 2006;6:204–216. - PubMed
-
- Lee AK, et al. Tumor marker expression in breast carcinomas and relationship to prognosis. An immunohistochemical study. Am J Clin Pathol. 1985;84:687–696. - PubMed
-
- Wrba F, et al. Prognostic significance of immunohistochemical parameters in breast carcinomas. Pathol Res Pract. 1988;183:277–283. - PubMed
-
- Cohen C, Sharkey FE, Shulman G, Uthman EO, Budgeon LR. Tumor-associated antigens in breast carcinomas. Prognostic significance. Cancer. 1987;60:1294–1298. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

