West Nile virus
- PMID: 20513541
- PMCID: PMC2905782
- DOI: 10.1016/j.cll.2009.10.006
West Nile virus
Abstract
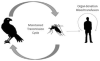
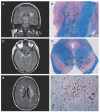
West Nile virus (WNV) is responsible for thousands of cases of morbidity and mortality in birds, horses, and humans. Epidemics were localized to Europe, Africa, the Middle East, and parts of Asia, and primarily caused a mild febrile illness in humans. In the late 1990s, the virus became more virulent and spread to North America. In humans, the clinical presentation ranges from asymptomatic, seen frequently, to encephalitis/paralysis and death, seen rarely. There is no FDA (Food and Drug Administration)-licensed vaccine for human use, and the only recommended treatment is supportive care. Often, there is a long recovery period. This article reviews the current literature summarizing the molecular virology, epidemiology, clinical manifestations, pathogenesis, diagnosis, treatment, immunology, and protective measures against WNV and WNV infections in humans.
2010 Elsevier Inc. All rights reserved.
Figures





References
-
- Gould EA, Solomon T. Pathogenic flaviviruses. Lancet. 2008;371(9611):500–9. - PubMed
-
- Lindenbach BD, Rice CM. Flaviviridae: the viruses and their replication. In: Knipe HP, editor. Fields Virology. Lippincott, Williams, & Wilkins; Philadelphia: 2001. pp. 991–1041. DM.
-
- Mackenzie JM, et al. Subcellular localization and some biochemical properties of the flavivirus Kunjin nonstructural proteins NS2A and NS4A. Virology. 1998;245(2):203–15. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

