Knock down of HIF-1alpha in glioma cells reduces migration in vitro and invasion in vivo and impairs their ability to form tumor spheres
- PMID: 20515450
- PMCID: PMC2896954
- DOI: 10.1186/1476-4598-9-133
Knock down of HIF-1alpha in glioma cells reduces migration in vitro and invasion in vivo and impairs their ability to form tumor spheres
Abstract
Background: Glioblastoma (GBM) is the most common and malignant primary intracranial human neoplasm. GBMs are characterized by the presence of extensive areas of necrosis and hypoxia. Hypoxia and its master regulator, hypoxia inducible factor 1 (HIF-1) play a key role in glioma invasion.
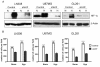
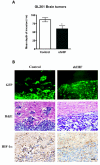
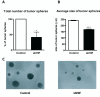
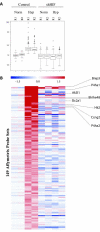
Results: To further elucidate the functional role of HIF-1alpha in glioma cell migration in vitro and in invasion in vivo, we used a shRNA approach to knock down HIF-1alpha expression complemented with genome-wide expression profiling, performed in both normoxic and hypoxic conditions. Our data show that knock down of HIF-1alpha in glioma cells significantly impairs their migration in vitro as well as their ability to invade into the brain parenchyma in vivo. Next, we assessed the role that HIF-1alpha plays in maintaining the characteristics of cancer stem cells (CSCs). By using the tumor sphere forming assay, we demonstrate that HIF-1alpha plays a role in the survival and self-renewal potential of CSCs. Finally, expression profiling experiments in glioma cells provided detailed insight into a broad range of specific biological pathways and processes downstream of HIF-1alpha. We discuss the role of these processes in the migratory and invasive properties, as well as the stem cell biology of glioblastomas
Conclusions: Our data show that knock down of HIF-1alpha in human and murine glioma cells impairs their migration in vitro and their invasion in vivo. In addition, our data suggest that HIF-1alpha plays a role in the survival and self-renewal potential of CSCs and identify genes that might further elucidate the role of HIF-1alpha in tumor migration, invasion and stem cell biology.
Figures
Similar articles
-
Silencing hypoxia-inducible factor-1alpha inhibits cell migration and invasion under hypoxic environment in malignant gliomas.Int J Oncol. 2007 Apr;30(4):793-802. Int J Oncol. 2007. PMID: 17332917
-
Geranylgeranyltransferase I regulates HIF-1α promoting glioblastoma cell migration and invasion.J Neurooncol. 2013 May;112(3):365-74. doi: 10.1007/s11060-013-1081-y. Epub 2013 Mar 9. J Neurooncol. 2013. PMID: 23475391
-
Suppressing the malignant phenotypes of glioma cells by lentiviral delivery of small hairpin RNA targeting hypoxia-inducible factor-1α.Int J Clin Exp Pathol. 2013 Oct 15;6(11):2323-32. eCollection 2013. Int J Clin Exp Pathol. 2013. PMID: 24228093 Free PMC article.
-
Developmentally regulated signaling pathways in glioma invasion.Cell Mol Life Sci. 2018 Feb;75(3):385-402. doi: 10.1007/s00018-017-2608-8. Epub 2017 Aug 18. Cell Mol Life Sci. 2018. PMID: 28821904 Free PMC article. Review.
-
Brain tumor stem cells as therapeutic targets in models of glioma.Yonsei Med J. 2010 Sep;51(5):633-40. doi: 10.3349/ymj.2010.51.5.633. Yonsei Med J. 2010. PMID: 20635435 Free PMC article. Review.
Cited by
-
Signal transduction molecule patterns indicating potential glioblastoma therapy approaches.Onco Targets Ther. 2013 Nov 29;6:1737-49. doi: 10.2147/OTT.S52365. eCollection 2013. Onco Targets Ther. 2013. PMID: 24348050 Free PMC article.
-
Antiangiogenic Potential of Microbial Metabolite Elaiophylin for Targeting Tumor Angiogenesis.Molecules. 2018 Mar 2;23(3):563. doi: 10.3390/molecules23030563. Molecules. 2018. PMID: 29498688 Free PMC article.
-
Antiangiogenic agents increase breast cancer stem cells via the generation of tumor hypoxia.Proc Natl Acad Sci U S A. 2012 Feb 21;109(8):2784-9. doi: 10.1073/pnas.1018866109. Epub 2012 Jan 23. Proc Natl Acad Sci U S A. 2012. PMID: 22308314 Free PMC article.
-
Molecular mechanisms for survival regulation of chronic myeloid leukemia stem cells.Protein Cell. 2013 Mar;4(3):186-96. doi: 10.1007/s13238-013-2115-0. Epub 2013 Mar 13. Protein Cell. 2013. PMID: 23483480 Free PMC article. Review.
-
The Role of Tumor Microenvironment in Regulating the Plasticity of Osteosarcoma Cells.Int J Mol Sci. 2022 Dec 18;23(24):16155. doi: 10.3390/ijms232416155. Int J Mol Sci. 2022. PMID: 36555795 Free PMC article. Review.
References
-
- Wu M, Chen Q, Li D, Li X, Li X, Huang C, Tang Y, Zhou Y, Wang D, Tang K. LRRC4 inhibits human glioblastoma cells proliferation, invasion, and proMMP-2 activation by reducing SDF-1 alpha/CXCR4-mediated ERK1/2 and Akt signaling pathways. J Cell Biochem. 2008;103:245–255. doi: 10.1002/jcb.21400. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

