Nitric oxide and superoxide transport in a cross section of the rat outer medulla. II. Reciprocal interactions and tubulovascular cross talk
- PMID: 20519375
- PMCID: PMC2944298
- DOI: 10.1152/ajprenal.00681.2009
Nitric oxide and superoxide transport in a cross section of the rat outer medulla. II. Reciprocal interactions and tubulovascular cross talk
Abstract
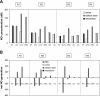
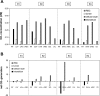
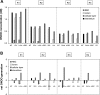
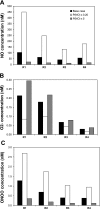
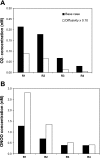
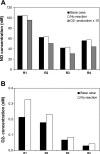
In a companion study (Edwards A and Layton AT. Am J Physiol Renal Physiol. doi:10.1152/ajprenal.00680.2009), we developed a mathematical model of nitric oxide (NO), superoxide (O(2)(-)), and total peroxynitrite (ONOO) transport in mid-outer stripe and mid-inner stripe cross sections of the rat outer medulla (OM). We examined how the three-dimensional architecture of the rat OM, together with low medullary oxygen tension (Po(2)), affects the distribution of NO, O(2)(-), and ONOO in the rat OM. In the current study, we sought to determine generation rate and permeability values that are compatible with measurements of medullary NO concentrations and to assess the importance of tubulovascular cross talk and NO-O(2)(-) interactions under physiological conditions. Our results suggest that the main determinants of NO concentrations in the rat OM are the rate of vascular and tubular NO synthesis under hypoxic conditions, and the red blood cell (RBC) permeability to NO (P(NO)(RBC)). The lower the P(NO)(RBC), the lower the amount of NO that is scavenged by hemoglobin species, and the higher the extra-erythrocyte NO concentrations. In addition, our results indicate that basal endothelial NO production acts to significantly limit NaCl reabsorption across medullary thick ascending limbs and to sustain medullary perfusion, whereas basal epithelial NO production has a smaller impact on NaCl transport and a negligible effect on vascular tone. Our model also predicts that O(2)(-) consumption by NO significantly reduces medullary O(2)(-) concentrations, but that O(2)(-) , when present at subnanomolar concentrations, has a small impact on medullary NO bioavailability.
Figures
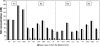
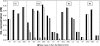

Similar articles
-
Nitric oxide and superoxide transport in a cross section of the rat outer medulla. I. Effects of low medullary oxygen tension.Am J Physiol Renal Physiol. 2010 Sep;299(3):F616-33. doi: 10.1152/ajprenal.00680.2009. Epub 2010 Jun 9. Am J Physiol Renal Physiol. 2010. PMID: 20534869 Free PMC article.
-
Modulation of outer medullary NaCl transport and oxygenation by nitric oxide and superoxide.Am J Physiol Renal Physiol. 2011 Nov;301(5):F979-96. doi: 10.1152/ajprenal.00096.2011. Epub 2011 Aug 17. Am J Physiol Renal Physiol. 2011. PMID: 21849492 Free PMC article.
-
Impact of nitric oxide-mediated vasodilation on outer medullary NaCl transport and oxygenation.Am J Physiol Renal Physiol. 2012 Oct;303(7):F907-17. doi: 10.1152/ajprenal.00055.2012. Epub 2012 Jul 11. Am J Physiol Renal Physiol. 2012. PMID: 22791340 Free PMC article.
-
Molecular mechanisms and therapeutic strategies of chronic renal injury: physiological role of angiotensin II-induced oxidative stress in renal medulla.J Pharmacol Sci. 2006 Jan;100(1):2-8. doi: 10.1254/jphs.fmj05003x2. Epub 2006 Jan 11. J Pharmacol Sci. 2006. PMID: 16404134 Review.
-
Nitric oxide and superoxide in the renal medulla: a delicate balancing act.Curr Opin Nephrol Hypertens. 2005 Jan;14(1):9-15. doi: 10.1097/00041552-200501000-00003. Curr Opin Nephrol Hypertens. 2005. PMID: 15586010 Review.
Cited by
-
Nitric oxide and superoxide transport in a cross section of the rat outer medulla. I. Effects of low medullary oxygen tension.Am J Physiol Renal Physiol. 2010 Sep;299(3):F616-33. doi: 10.1152/ajprenal.00680.2009. Epub 2010 Jun 9. Am J Physiol Renal Physiol. 2010. PMID: 20534869 Free PMC article.
-
Modulation of outer medullary NaCl transport and oxygenation by nitric oxide and superoxide.Am J Physiol Renal Physiol. 2011 Nov;301(5):F979-96. doi: 10.1152/ajprenal.00096.2011. Epub 2011 Aug 17. Am J Physiol Renal Physiol. 2011. PMID: 21849492 Free PMC article.
-
A new microscope for the kidney: mathematics.Am J Physiol Renal Physiol. 2017 Apr 1;312(4):F671-F672. doi: 10.1152/ajprenal.00648.2016. Epub 2017 Jan 18. Am J Physiol Renal Physiol. 2017. PMID: 28100504 Free PMC article. No abstract available.
-
Modeling Transport and Flow Regulatory Mechanisms of the Kidney.ISRN Biomath. 2012 Jul 12;2012(2012):170594. doi: 10.5402/2012/170594. ISRN Biomath. 2012. PMID: 23914303 Free PMC article.
-
Cellular mechanisms underlying nitric oxide-induced vasodilation of descending vasa recta.Am J Physiol Renal Physiol. 2011 Feb;300(2):F441-56. doi: 10.1152/ajprenal.00499.2010. Epub 2010 Nov 17. Am J Physiol Renal Physiol. 2011. PMID: 21084408 Free PMC article.
References
-
- Abe M, O'Connor P, Kaldunski M, Liang M, Roman RJ, Cowley AW., Jr Effect of sodium delivery on superoxide and nitric oxide in the medullary thick ascending limb. Am J Physiol Renal Physiol 291: F350–F357, 2006 - PubMed
-
- Buerk D, Lamkin-Kennard K, Jaron D. Modeling the influence of superoxide dismutase on superoxide and nitric oxide interactions, including reversible inhibition of oxygen consumption. Free Radic Biol Med 34: 1488–1503, 2003 - PubMed
-
- Cassoly R, Gibson QH. Conformation, co-operativity and ligand binding in human hemoglobin. J Mol Biol 91: 301–313, 1975. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

