Selective changes in thin spine density and morphology in monkey prefrontal cortex correlate with aging-related cognitive impairment
- PMID: 20519525
- PMCID: PMC2892969
- DOI: 10.1523/JNEUROSCI.6410-09.2010
Selective changes in thin spine density and morphology in monkey prefrontal cortex correlate with aging-related cognitive impairment
Abstract
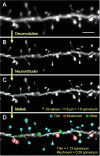
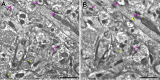
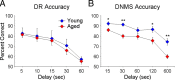
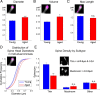
Age-associated memory impairment (AAMI) occurs in many mammalian species, including humans. In contrast to Alzheimer's disease (AD), in which circuit disruption occurs through neuron death, AAMI is due to circuit and synapse disruption in the absence of significant neuron loss and thus may be more amenable to prevention or treatment. We have investigated the effects of aging on pyramidal neurons and synapse density in layer III of area 46 in dorsolateral prefrontal cortex of young and aged, male and female rhesus monkeys (Macaca mulatta) that were tested for cognitive status through the delayed non-matching-to-sample (DNMS) and delayed response tasks. Cognitive tests revealed an age-related decrement in both acquisition and performance on DNMS. Our morphometric analyses revealed both an age-related loss of spines (33%, p < 0.05) on pyramidal cells and decreased density of axospinous synapses (32%, p < 0.01) in layer III of area 46. In addition, there was an age-related shift in the distribution of spine types reflecting a selective vulnerability of small, thin spines, thought to be particularly plastic and linked to learning. While both synapse density and the overall spine size average of an animal were predictive of number of trials required for acquisition of DNMS (i.e., learning the task), the strongest correlate of behavior was found to be the head volume of thin spines, with no correlation between behavior and mushroom spine size or density. No synaptic index correlated with memory performance once the task was learned.
Figures




References
-
- Bachevalier J, Landis LS, Walker LC, Brickson M, Mishkin M, Price DL, Cork LC. Aged monkeys exhibit behavioral deficits indicative of widespread cerebral dysfunction. Neurobiol Aging. 1991;12:99–111. - PubMed
-
- Baxter MG, Murray EA. Opposite relationship of hippocampal and rhinal cortex damage to delayed nonmatching-to-sample deficits in monkeys. Hippocampus. 2001;11:61–71. - PubMed
-
- Beason-Held LL, Rosene DL, Killiany RJ, Moss MB. Hippocampal formation lesions produce memory impairment in the rhesus monkey. Hippocampus. 1999;9:562–574. - PubMed
-
- Bourne J, Harris KM. Do thin spines learn to be mushroom spines that remember? Curr Opin Neurobiol. 2007;17:381–386. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
