YopD self-assembly and binding to LcrV facilitate type III secretion activity by Yersinia pseudotuberculosis
- PMID: 20525687
- PMCID: PMC2919090
- DOI: 10.1074/jbc.M110.144311
YopD self-assembly and binding to LcrV facilitate type III secretion activity by Yersinia pseudotuberculosis
Abstract
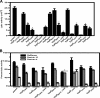
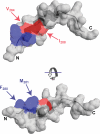
YopD-like translocator proteins encoded by several Gram-negative bacteria are important for type III secretion-dependent delivery of anti-host effectors into eukaryotic cells. This probably depends on their ability to form pores in the infected cell plasma membrane, through which effectors may gain access to the cell interior. In addition, Yersinia YopD is a negative regulator essential for the control of effector synthesis and secretion. As a prerequisite for this functional duality, YopD may need to establish molecular interactions with other key T3S components. A putative coiled-coil domain and an alpha-helical amphipathic domain, both situated in the YopD C terminus, may represent key protein-protein interaction domains. Therefore, residues within the YopD C terminus were systematically mutagenized. All 68 mutant bacteria were first screened in a variety of assays designed to identify individual residues essential for YopD function, possibly by providing the interaction interface for the docking of other T3S proteins. Mirroring the effect of a full-length yopD gene deletion, five mutant bacteria were defective for both yop regulatory control and effector delivery. Interestingly, all mutations clustered to hydrophobic amino acids of the amphipathic domain. Also situated within this domain, two additional mutants rendered YopD primarily defective in the control of Yop synthesis and secretion. Significantly, protein-protein interaction studies revealed that functionally compromised YopD variants were also defective in self-oligomerization and in the ability to engage another translocator protein, LcrV. Thus, the YopD amphipathic domain facilitates the formation of YopD/YopD and YopD/LcrV interactions, two critical events in the type III secretion process.
Figures





Similar articles
-
A mutant with aberrant extracellular LcrV-YscF interactions fails to form pores and translocate Yop effector proteins but retains the ability to trigger Yop secretion in response to host cell contact.J Bacteriol. 2013 May;195(10):2244-54. doi: 10.1128/JB.02011-12. Epub 2013 Mar 8. J Bacteriol. 2013. PMID: 23475976 Free PMC article.
-
YopD of Yersinia pseudotuberculosis is translocated into the cytosol of HeLa epithelial cells: evidence of a structural domain necessary for translocation.Mol Microbiol. 1998 Aug;29(3):799-813. doi: 10.1046/j.1365-2958.1998.00973.x. Mol Microbiol. 1998. PMID: 9723919
-
Yersinia pseudotuberculosis YopD mutants that genetically separate effector protein translocation from host membrane disruption.Mol Microbiol. 2015 May;96(4):764-78. doi: 10.1111/mmi.12970. Epub 2015 Mar 16. Mol Microbiol. 2015. PMID: 25684661
-
The Yersinia Yop virulon: a bacterial system for subverting eukaryotic cells.Mol Microbiol. 1997 Mar;23(5):861-7. doi: 10.1046/j.1365-2958.1997.2731623.x. Mol Microbiol. 1997. PMID: 9076724 Review.
-
Regulation of the Yersinia type III secretion system: traffic control.Front Cell Infect Microbiol. 2013 Feb 6;3:4. doi: 10.3389/fcimb.2013.00004. eCollection 2013. Front Cell Infect Microbiol. 2013. PMID: 23390616 Free PMC article. Review.
Cited by
-
Translational regulation of Yersinia enterocolitica mRNA encoding a type III secretion substrate.J Biol Chem. 2013 Dec 6;288(49):35478-88. doi: 10.1074/jbc.M113.504811. Epub 2013 Oct 24. J Biol Chem. 2013. PMID: 24158443 Free PMC article.
-
The Type III Secretion Translocation Pore Senses Host Cell Contact.PLoS Pathog. 2016 Mar 29;12(3):e1005530. doi: 10.1371/journal.ppat.1005530. eCollection 2016 Mar. PLoS Pathog. 2016. PMID: 27022930 Free PMC article.
-
YspC: a unique translocator exhibits structural alteration in the complex form with chaperone SycB.Protein J. 2012 Aug;31(6):487-98. doi: 10.1007/s10930-012-9426-5. Protein J. 2012. PMID: 22648693
-
Distinct mechanisms of type 3 secretion system recognition control LTB4 synthesis in neutrophils and macrophages.PLoS Pathog. 2024 Oct 18;20(10):e1012651. doi: 10.1371/journal.ppat.1012651. eCollection 2024 Oct. PLoS Pathog. 2024. PMID: 39423229 Free PMC article.
-
Delivering the pain: an overview of the type III secretion system with special consideration for aquatic pathogens.Vet Res. 2021 Dec 19;52(1):146. doi: 10.1186/s13567-021-01015-8. Vet Res. 2021. PMID: 34924019 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

