Palmitate induced IL-6 and MCP-1 expression in human bladder smooth muscle cells provides a link between diabetes and urinary tract infections
- PMID: 20526368
- PMCID: PMC2878332
- DOI: 10.1371/journal.pone.0010882
Palmitate induced IL-6 and MCP-1 expression in human bladder smooth muscle cells provides a link between diabetes and urinary tract infections
Abstract
Background: Urinary tract infections (UTI) are more frequent in type-2 diabetes mellitus patients than in subjects with normal glucose metabolism. The mechanisms underlying this higher prevalence of UTI are unknown. However, cytokine levels are altered in diabetic patients and may thus contribute to the development of UTI. Increased levels of free fatty acids (FFA), as observed in obese patients, can induce IL-6 production in various cell types. Therefore we studied the effects of the free fatty acid palmitate and bacterial lipopolysaccharide (LPS) on interleukin-6 (IL-6) and monocyte chemotactic protein-1 (MCP-1) expression and secretion in cultured human bladder smooth muscle cells (hBSMC).
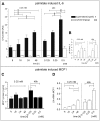
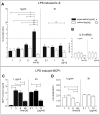
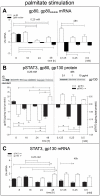
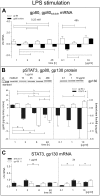
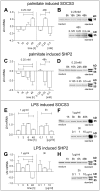
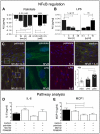
Methodology/principal findings: Biopsies were taken from patients undergoing cystectomy due to bladder cancer. Palmitate or LPS stimulated hBSMC were analysed for the production and secretion of the IL-6, gp80, gp80soluble, gp130, MCP-1, pSTAT3, SOCS3, NF-kappaB and SHP2 by quantitative PCR, ELISA, Western blotting, and confocal immunofluorescence. In signal transduction inhibition experiments we evaluated the involvement of NF-kappaB and MEK1 in IL-6 and MCP-1 regulation. Palmitate upregulates IL-6 mRNA expression and secretion via NF-kappaB dependent pathways in a concentration- and time-dependent manner. MCP-1 was moderately upregulated by palmitate but was strongly upregulated by LPS involving NF-kappaB and MEK1 dependent pathways. Soluble IL-6 receptor (gp80soluble) was downregulated by palmitate and LPS, while membrane-bound gp80 was moderately upregulated. LPS increased SOCS3 and SHP2, whereas palmitate only induced SOCS3. Secondary finding: most of the IL-6 is secreted.
Conclusions/significance: Bacterial infection (LPS) or metabolic alterations (palmitate) have distinct effects on IL-6 expression in hBSMC, (i) short term LPS induced autocrine JAK/STAT signaling and (ii) long-term endocrine regulation of IL-6 by palmitate. Induction of IL-6 in human bladder smooth muscle cells by fatty acids may represent a pathogenetic factor underlying the higher frequency and persistence of urinary tract infections in patients with metabolic diseases.
Conflict of interest statement
Figures

References
-
- Benfield T, Jensen JS, Nordestgaard BG. Influence of diabetes and hyperglycaemia on infectious disease hospitalisation and outcome. Diabetologia. 2007;50:549–554. - PubMed
-
- Stapleton A. Urinary tract infections in patients with diabetes. Am J Med. 2002;113(Suppl 1A):80S–84S. - PubMed
-
- Hoepelman AI, Meiland R, Geerlings SE. Pathogenesis and management of bacterial urinary tract infections in adult patients with diabetes mellitus. Int J Antimicrob Agents. 2003;22(Suppl 2):35–43. - PubMed
-
- Harding GK, Zhanel GG, Nicolle LE, Cheang M. Antimicrobial treatment in diabetic women with asymptomatic bacteriuria. N Engl J Med. 2002;347:1576–1583. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

