Methylthioadenosine (MTA) inhibits melanoma cell proliferation and in vivo tumor growth
- PMID: 20529342
- PMCID: PMC2891639
- DOI: 10.1186/1471-2407-10-265
Methylthioadenosine (MTA) inhibits melanoma cell proliferation and in vivo tumor growth
Abstract
Background: Melanoma is the most deadly form of skin cancer without effective treatment. Methylthioadenosine (MTA) is a naturally occurring nucleoside with differential effects on normal and transformed cells. MTA has been widely demonstrated to promote anti-proliferative and pro-apoptotic responses in different cell types. In this study we have assessed the therapeutic potential of MTA in melanoma treatment.
Methods: To investigate the therapeutic potential of MTA we performed in vitro proliferation and viability assays using six different mouse and human melanoma cell lines wild type for RAS and BRAF or harboring different mutations in RAS pathway. We also have tested its therapeutic capabilities in vivo in a xenograft mouse melanoma model and using variety of molecular techniques and tissue culture we investigated its anti-proliferative and pro-apoptotic properties.
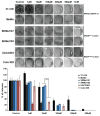
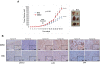
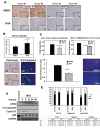
Results: In vitro experiments showed that MTA treatment inhibited melanoma cell proliferation and viability in a dose dependent manner, where BRAF mutant melanoma cell lines appear to be more sensitive. Importantly, MTA was effective inhibiting in vivo tumor growth. The molecular analysis of tumor samples and in vitro experiments indicated that MTA induces cytostatic rather than pro-apoptotic effects inhibiting the phosphorylation of Akt and S6 ribosomal protein and inducing the down-regulation of cyclin D1.
Conclusions: MTA inhibits melanoma cell proliferation and in vivo tumor growth particularly in BRAF mutant melanoma cells. These data reveal a naturally occurring drug potentially useful for melanoma treatment.
Figures

References
-
- Savarese TM, Ghoda LY, Dexter DL, Parks RE Jr. Conversion of 5'-deoxy-5'-methylthioadenosine and 5'-deoxy-5'-methylthioinosine to methionine in cultured human leukemic cells. Cancer Res. 1983;43(10):4699–4702. - PubMed
-
- Olopade OI, Pomykala HM, Hagos F, Sveen LW, Espinosa R, Dreyling MH, Gursky S, Stadler WM, Le Beau MM, Bohlander SK. Construction of a 2.8-megabase yeast artificial chromosome contig and cloning of the human methylthioadenosine phosphorylase gene from the tumor suppressor region on 9p21. Proc Natl Acad Sci USA. 1995;92(14):6489–6493. doi: 10.1073/pnas.92.14.6489. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

