Random X inactivation and extensive mosaicism in human placenta revealed by analysis of allele-specific gene expression along the X chromosome
- PMID: 20532033
- PMCID: PMC2881032
- DOI: 10.1371/journal.pone.0010947
Random X inactivation and extensive mosaicism in human placenta revealed by analysis of allele-specific gene expression along the X chromosome
Abstract
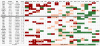
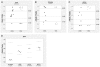
Imprinted inactivation of the paternal X chromosome in marsupials is the primordial mechanism of dosage compensation for X-linked genes between females and males in Therians. In Eutherian mammals, X chromosome inactivation (XCI) evolved into a random process in cells from the embryo proper, where either the maternal or paternal X can be inactivated. However, species like mouse and bovine maintained imprinted XCI exclusively in extraembryonic tissues. The existence of imprinted XCI in humans remains controversial, with studies based on the analyses of only one or two X-linked genes in different extraembryonic tissues. Here we readdress this issue in human term placenta by performing a robust analysis of allele-specific expression of 22 X-linked genes, including XIST, using 27 SNPs in transcribed regions. We show that XCI is random in human placenta, and that this organ is arranged in relatively large patches of cells with either maternal or paternal inactive X. In addition, this analysis indicated heterogeneous maintenance of gene silencing along the inactive X, which combined with the extensive mosaicism found in placenta, can explain the lack of agreement among previous studies. Our results illustrate the differences of XCI mechanism between humans and mice, and highlight the importance of addressing the issue of imprinted XCI in other species in order to understand the evolution of dosage compensation in placental mammals.
Conflict of interest statement
Figures
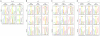

Similar articles
-
No imprinted XIST expression in pigs: biallelic XIST expression in early embryos and random X inactivation in placentas.Cell Mol Life Sci. 2019 Nov;76(22):4525-4538. doi: 10.1007/s00018-019-03123-3. Epub 2019 May 28. Cell Mol Life Sci. 2019. PMID: 31139846 Free PMC article.
-
Regulation of X-chromosome inactivation in development in mice and humans.Microbiol Mol Biol Rev. 1998 Jun;62(2):362-78. doi: 10.1128/MMBR.62.2.362-378.1998. Microbiol Mol Biol Rev. 1998. PMID: 9618446 Free PMC article. Review.
-
Eutherian mammals use diverse strategies to initiate X-chromosome inactivation during development.Nature. 2011 Apr 21;472(7343):370-4. doi: 10.1038/nature09872. Epub 2011 Apr 6. Nature. 2011. PMID: 21471966
-
Random X inactivation in the mule and horse placenta.Genome Res. 2012 Oct;22(10):1855-63. doi: 10.1101/gr.138487.112. Epub 2012 May 29. Genome Res. 2012. PMID: 22645258 Free PMC article.
-
Imprinted X inactivation in eutherians: a model of gametic execution and zygotic relaxation.Curr Opin Cell Biol. 2001 Dec;13(6):690-7. doi: 10.1016/s0955-0674(00)00272-6. Curr Opin Cell Biol. 2001. PMID: 11698184 Review.
Cited by
-
Mechanisms of Choice in X-Chromosome Inactivation.Cells. 2022 Feb 3;11(3):535. doi: 10.3390/cells11030535. Cells. 2022. PMID: 35159344 Free PMC article. Review.
-
The Placenta's Role in Sexually Dimorphic Fetal Growth Strategies.Reprod Sci. 2022 Jun;29(6):1895-1907. doi: 10.1007/s43032-021-00780-3. Epub 2021 Oct 26. Reprod Sci. 2022. PMID: 34699045 Review.
-
Sex Differences Are Here to Stay: Relevance to Prenatal Care.J Clin Med. 2021 Jul 5;10(13):3000. doi: 10.3390/jcm10133000. J Clin Med. 2021. PMID: 34279482 Free PMC article. Review.
-
Changes in gene expression following long-term in vitro exposure of Macaca mulatta trophoblast stem cells to biologically relevant levels of endocrine disruptors.Reprod Toxicol. 2018 Apr;77:154-165. doi: 10.1016/j.reprotox.2018.02.012. Epub 2018 Mar 2. Reprod Toxicol. 2018. PMID: 29505797 Free PMC article.
-
Single-Cell RNA-Seq Reveals Lineage and X Chromosome Dynamics in Human Preimplantation Embryos.Cell. 2016 May 5;165(4):1012-26. doi: 10.1016/j.cell.2016.03.023. Epub 2016 Apr 7. Cell. 2016. PMID: 27062923 Free PMC article.
References
-
- Cooper DW, VandeBerg JL, Sharman GB, Poole WE. Phosphoglycerate kinase polymorphism in kangaroos provides further evidence for paternal X inactivation. Nat New Biol. 1971;230:155–157. - PubMed
-
- Sharman GB. Late DNA replication in the paternally derived X chromosome of female kangaroos. Nature. 1971;230:231–232. - PubMed
-
- Takagi N, Sasaki M. Preferential inactivation of the paternally derived X chromosome in the extraembryonic membranes of the mouse. Nature. 1975;256:640–642. - PubMed
-
- West JD, Frels WI, Chapman VM, Papaioannou VE. Preferential expression of the maternally derived X chromosome in the mouse yolk sac. Cell. 1977;12:873–882. - PubMed
-
- Wake N, Takagi N, Sasaki M. Non-random inactivation of X chromosome in the rat yolk sac. Nature. 1976;262:580–581. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

