Defensin-like ZmES4 mediates pollen tube burst in maize via opening of the potassium channel KZM1
- PMID: 20532241
- PMCID: PMC2879413
- DOI: 10.1371/journal.pbio.1000388
Defensin-like ZmES4 mediates pollen tube burst in maize via opening of the potassium channel KZM1
Abstract
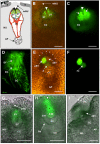
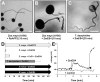
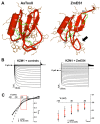
In contrast to animals and lower plant species, sperm cells of flowering plants are non-motile and are transported to the female gametes via the pollen tube, i.e. the male gametophyte. Upon arrival at the female gametophyte two sperm cells are discharged into the receptive synergid cell to execute double fertilization. The first players involved in inter-gametophyte signaling to attract pollen tubes and to arrest their growth have been recently identified. In contrast the physiological mechanisms leading to pollen tube burst and thus sperm discharge remained elusive. Here, we describe the role of polymorphic defensin-like cysteine-rich proteins ZmES1-4 (Zea mays embryo sac) from maize, leading to pollen tube growth arrest, burst, and explosive sperm release. ZmES1-4 genes are exclusively expressed in the cells of the female gametophyte. ZmES4-GFP fusion proteins accumulate in vesicles at the secretory zone of mature synergid cells and are released during the fertilization process. Using RNAi knock-down and synthetic ZmES4 proteins, we found that ZmES4 induces pollen tube burst in a species-preferential manner. Pollen tube plasma membrane depolarization, which occurs immediately after ZmES4 application, as well as channel blocker experiments point to a role of K(+)-influx in the pollen tube rupture mechanism. Finally, we discovered the intrinsic rectifying K(+) channel KZM1 as a direct target of ZmES4. Following ZmES4 application, KZM1 opens at physiological membrane potentials and closes after wash-out. In conclusion, we suggest that vesicles containing ZmES4 are released from the synergid cells upon male-female gametophyte signaling. Subsequent interaction between ZmES4 and KZM1 results in channel opening and K(+) influx. We further suggest that K(+) influx leads to water uptake and culminates in osmotic tube burst. The species-preferential activity of polymorphic ZmES4 indicates that the mechanism described represents a pre-zygotic hybridization barrier and may be a component of reproductive isolation in plants.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

References
-
- Bell C. D, Soltis D. E, Soltis P. S. The age of the angiosperms: a molecular timescale without a clock. Evolution. 2005;59:1245–1258. - PubMed
-
- Punwani J. A, Drews G. N. Development and function of the synergid cell. Sex Plant Reprod. 2008;21:7–15.
-
- Dresselhaus T. Cell-cell communication during double fertilization. Curr Opin Plant Biol. 2006;9:41–47. - PubMed
-
- Strasburger E. Neuere untersuchungen über den befruchtungsvorgang bei den phanerogamen als grundlage für eine theorie der zeugung. Jena, Gustav Fischer 1884
-
- Escobar-Restrepo J. M, Huck N, Kessler S, Gagliardini V, Gheyselinck J, et al. The FERONIA receptor-like kinase mediates male-female interactions during pollen tube reception. Science. 2007;317:656–660. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

