Continuous elevation of PTH increases the number of osteoblasts via both osteoclast-dependent and -independent mechanisms
- PMID: 20533302
- PMCID: PMC3179285
- DOI: 10.1002/jbmr.145
Continuous elevation of PTH increases the number of osteoblasts via both osteoclast-dependent and -independent mechanisms
Abstract
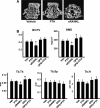
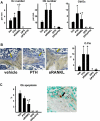
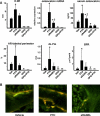
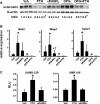
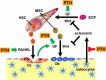
Sustained parathyroid hormone (PTH) elevation stimulates bone remodeling (ie, both resorption and formation). The former results from increased RANKL synthesis, but the cause of the latter has not been established. Current hypotheses include release of osteoblastogenic factors from osteoclasts or from the bone matrix during resorption, modulation of the production and activity of osteoblastogenic factors from cells of the osteoblast lineage, and increased angiogenesis. To dissect the contribution of these mechanisms, 6-month-old Swiss-Webster mice were infused for 5 days with 470 ng/h PTH(1-84) or 525 ng/h soluble RANKL (sRANKL). Both agents increased osteoclasts and osteoblasts in vertebral cancellous bone, but the ratio of osteoblasts to osteoclasts and the increase in bone formation was greater in PTH-treated mice. Cancellous bone mass was maintained in mice receiving PTH but lost in mice receiving sRANKL, indicating that maintenance of balanced remodeling requires osteoblastogenic effects beyond those mediated by osteoclasts. Consistent with this contention, PTH, but not sRANKL, decreased the level of the Wnt antagonist sclerostin and increased the expression of the Wnt target genes Nkd2, Wisp1, and Twist1. Furthermore, PTH, but not sRANKL, increased the number of blood vessels in the bone marrow. Weekly injections of the RANKL antagonist osteoprotegerin at 10 µg/g for 2 weeks prior to PTH infusion eliminated osteoclasts and osteoblasts and prevented the PTH-induced increase in osteoclasts, osteoblasts, and blood vessels. These results indicate that PTH stimulates osteoclast-dependent as well as osteoclast-independent (Wnt signaling) pro-osteoblastogenic pathways, both of which are required for balanced focal bone remodeling in cancellous bone.
© 2010 American Society for Bone and Mineral Research.
Figures


References
-
- Hauge EM, Qvesel D, Eriksen EF, Mosekilde L, Melsen F. Cancellous bone remodeling occurs in specialized compartments lined by cells expressing osteoblastic markers. J Bone Miner Res. 2001;16:1575–1582. - PubMed
-
- Parisien M, Dempster DW, Shane E, Bilezikian JP. Histomorphometric analysis of bone in primary hyperparathyroidism. In: Bilezikian JP, Marcus R, Levine MA, editors. The Parathyroids. Basic and Clinical Concepts. San Diego, CA: Academic Press; 2001. pp. 423–436.
-
- Mosekilde L. Primary hyperparathyroidism and the skeleton. Clin Endocrinol (Oxf). 2008;69:1–19. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

