Enhancement of intervertebral disc cell senescence by WNT/β-catenin signaling-induced matrix metalloproteinase expression
- PMID: 20533544
- PMCID: PMC3622204
- DOI: 10.1002/art.27599
Enhancement of intervertebral disc cell senescence by WNT/β-catenin signaling-induced matrix metalloproteinase expression
Abstract
Objective: To determine whether intervertebral disc (IVD) cells express β-catenin and to assess the role of the WNT/β-catenin signaling pathway in cellular senescence and aggrecan synthesis.
Methods: The expression of β-catenin messenger RNA (mRNA) and protein in rat IVD cells was assessed by using several real-time reverse transcription-polymerase chain reaction, Western blot, immunohistochemical, and immunofluorescence analyses. The effect of WNT/β-catenin on nucleus pulposus (NP) cells was examined by transfection experiments, an MTT assay, senescence-associated β-galactosidase staining, a cell cycle analysis, and a transforming growth factor (TGFβ)/bone morphogenetic protein (BMP) pathway-focused microarray analysis.
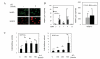
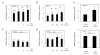
Results: We found that β-catenin mRNA and protein were expressed in discs in vivo and that rat NP cells exhibited increased β-catenin mRNA and protein upon stimulation with lithium chloride, a known activator of WNT signaling. LiCl treatment inhibited the proliferation of NP cells in a time- and dose-dependent manner. In addition, there was an increased level of cellular senescence in LiCl-treated cells. Long-term treatment with LiCl induced cell cycle arrest and promoted subsequent apoptosis in NP cells. Activation of WNT/β-catenin signaling also regulated the expression of aggrecan. We also demonstrated that WNT/β-catenin signaling induced the expression of matrix metalloproteinases (MMPs) and TGFβ in NP cells.
Conclusion: The activation of WNT/β-catenin signaling promotes cellular senescence and may modulate MMP and TGFβ signaling in NP cells. We hypothesize that the activation of WNT/β-catenin signaling may lead to an increased breakdown of the matrix, thereby promoting IVD degeneration.
Figures


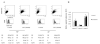

Similar articles
-
CCN family member 2/connective tissue growth factor (CCN2/CTGF) is regulated by Wnt-β-catenin signaling in nucleus pulposus cells.Arthritis Res Ther. 2018 Sep 29;20(1):217. doi: 10.1186/s13075-018-1723-8. Arthritis Res Ther. 2018. PMID: 30268161 Free PMC article.
-
The relationship between the Wnt/β-catenin and TGF-β/BMP signals in the intervertebral disc cell.J Cell Physiol. 2011 May;226(5):1139-48. doi: 10.1002/jcp.22438. J Cell Physiol. 2011. PMID: 20945354
-
Protein Kinase C and Matrix Metalloproteinases Expression Using Phorbol Myristate Acetate in Degenerative Intervertebral Disc Cells.Clin Orthop Surg. 2024 Oct;16(5):827-835. doi: 10.4055/cios23365. Epub 2024 Sep 13. Clin Orthop Surg. 2024. PMID: 39364116 Free PMC article.
-
Role of the Wnt pathway in the formation, development, and degeneration of intervertebral discs.Pathol Res Pract. 2021 Apr;220:153366. doi: 10.1016/j.prp.2021.153366. Epub 2021 Feb 8. Pathol Res Pract. 2021. PMID: 33647863 Review.
-
[Research progress of Wnt/beta-catenin and nuclear factor-kappa B pathways and their relevance to intervertebral disc degeneration].Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2013 Dec;27(12):1523-8. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2013. PMID: 24640378 Review. Chinese.
Cited by
-
IL-6/YAP1/β-catenin signaling is involved in intervertebral disc degeneration.J Cell Physiol. 2019 May;234(5):5964-5971. doi: 10.1002/jcp.27065. Epub 2018 Dec 3. J Cell Physiol. 2019. PMID: 30511395 Free PMC article.
-
Specific inhibitory protein Dkk-1 blocking Wnt/β-catenin signaling pathway improve protectives effect on the extracellular matrix.J Huazhong Univ Sci Technolog Med Sci. 2011 Oct;31(5):657. doi: 10.1007/s11596-011-0577-y. Epub 2011 Oct 25. J Huazhong Univ Sci Technolog Med Sci. 2011. PMID: 22038356
-
Mesenchymal stem cells-based therapy as a potential treatment in neurodegenerative disorders: is the escape from senescence an answer?Neural Regen Res. 2015 Jun;10(6):850-8. doi: 10.4103/1673-5374.158352. Neural Regen Res. 2015. PMID: 26199588 Free PMC article. Review.
-
High Glucose-Induced Oxidative Stress Mediates Apoptosis and Extracellular Matrix Metabolic Imbalances Possibly via p38 MAPK Activation in Rat Nucleus Pulposus Cells.J Diabetes Res. 2016;2016:3765173. doi: 10.1155/2016/3765173. Epub 2016 Aug 22. J Diabetes Res. 2016. PMID: 27635402 Free PMC article.
-
A role for TNFα in intervertebral disc degeneration: a non-recoverable catabolic shift.Biochem Biophys Res Commun. 2013 Mar 29;433(1):151-6. doi: 10.1016/j.bbrc.2013.02.034. Epub 2013 Feb 22. Biochem Biophys Res Commun. 2013. PMID: 23438440 Free PMC article.
References
-
- Sakai D, Mochida J, Yamamoto Y, Nomura T, Okuma M, Nishimura K, et al. Transplantation of mesenchymal stem cells embedded in Atelocollagen gel to the intervertebral disc: a potential therapeutic model for disc degeneration. Biomaterials. 2003;24:3531–41. - PubMed
-
- Masuda K, Oegema T, An H. Growth factors and treatment of intervertebral disc degeneration. Spine. 2004;29:2757–69. - PubMed
-
- Tropiano P, Huang RC, Girardi FP, Marnay T. Lumbar disc replacement: preliminary results with ProDisc II after a minimum follow-up period of 1 year. J Spinal Disord Tech. 2003;16:362–8. - PubMed
-
- Thompson JP, Oegema TR, Jr, Bradford DS. Stimulation of mature canine intervertebral disc by growth factors. Spine. 1991;16:253–60. - PubMed
-
- Hiyama A, Mochida J, Iwashina T, Omi H, Watanabe T, Serigano K, et al. Transplantation of mesenchymal stem cells in a canine disc degeneration model. J Orthop Res. 2008;26:589–600. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

