CXCR4 blockade augments bone marrow progenitor cell recruitment to the neovasculature and reduces mortality after myocardial infarction
- PMID: 20534467
- PMCID: PMC2890743
- DOI: 10.1073/pnas.0914248107
CXCR4 blockade augments bone marrow progenitor cell recruitment to the neovasculature and reduces mortality after myocardial infarction
Abstract
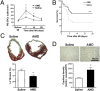
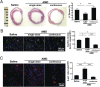
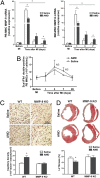
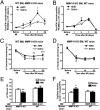
We hypothesized that a small molecule CXCR4 antagonist, AMD3100 (AMD), could augment the mobilization of bone marrow (BM)-derived endothelial progenitor cells (EPCs), thereby enhancing neovascularization and functional recovery after myocardial infarction. Single-dose AMD injection administered after the onset of myocardial infarction increased circulating EPC counts and myocardial vascularity, reduced fibrosis, and improved cardiac function and survival. In mice transplanted with traceable BM cells, AMD increased BM-derived cell incorporation in the ischemic border zone. In contrast, continuous infusion of AMD, although increasing EPCs in the circulation, worsened outcome by blocking EPC incorporation. In addition to its effects as a CXCR4 antagonist, AMD also up-regulated VEGF and matrix metalloproteinase 9 (MMP-9) expression, and the benefits of AMD were not observed in the absence of MMP-9 expression in the BM. These findings suggest that AMD3100 preserves cardiac function after myocardial infarction by enhancing BM-EPC-mediated neovascularization, and that these benefits require MMP-9 expression in the BM, but not in the ischemic region. Our results indicate that AMD3100 could be a potentially useful therapy for the treatment of myocardial infarction.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
CXC-chemokine receptor 4 antagonist AMD3100 promotes cardiac functional recovery after ischemia/reperfusion injury via endothelial nitric oxide synthase-dependent mechanism.Circulation. 2013 Jan 1;127(1):63-73. doi: 10.1161/CIRCULATIONAHA.112.099242. Epub 2012 Nov 30. Circulation. 2013. PMID: 23204107 Free PMC article.
-
Estradiol enhances recovery after myocardial infarction by augmenting incorporation of bone marrow-derived endothelial progenitor cells into sites of ischemia-induced neovascularization via endothelial nitric oxide synthase-mediated activation of matrix metalloproteinase-9.Circulation. 2006 Mar 28;113(12):1605-14. doi: 10.1161/CIRCULATIONAHA.105.553925. Epub 2006 Mar 13. Circulation. 2006. PMID: 16534014
-
Sonic hedgehog-induced functional recovery after myocardial infarction is enhanced by AMD3100-mediated progenitor-cell mobilization.J Am Coll Cardiol. 2011 Jun 14;57(24):2444-52. doi: 10.1016/j.jacc.2010.11.069. J Am Coll Cardiol. 2011. PMID: 21658566 Free PMC article.
-
Hypoxia inducible factor-1 alpha, endothelial progenitor cells, monocytes, cardiovascular risk, wound healing, cobalt and hydralazine: a unifying hypothesis.Curr Drug Targets. 2008 May;9(5):422-35. doi: 10.2174/138945008784221215. Curr Drug Targets. 2008. PMID: 18473772 Review.
-
Cell therapy with bone marrow cells for myocardial regeneration.Antioxid Redox Signal. 2009 Aug;11(8):1897-911. doi: 10.1089/ars.2009.2486. Antioxid Redox Signal. 2009. PMID: 19203213 Free PMC article. Review.
Cited by
-
Recent concepts for the roles of progenitor/stem cell niche in heart repair.Am J Cardiovasc Dis. 2012;2(1):75-83. Epub 2011 Dec 15. Am J Cardiovasc Dis. 2012. PMID: 22254217 Free PMC article.
-
Anti-inflammatory therapies in myocardial infarction: failures, hopes and challenges.Br J Pharmacol. 2018 May;175(9):1377-1400. doi: 10.1111/bph.14155. Epub 2018 Mar 4. Br J Pharmacol. 2018. PMID: 29394499 Free PMC article. Review.
-
CXCR4 signaling at the ovine fetal-maternal interface regulates vascularization, CD34+ cell presence, and autophagy in the endometrium†.Biol Reprod. 2019 Jul 1;101(1):102-111. doi: 10.1093/biolre/ioz073. Biol Reprod. 2019. PMID: 31004477 Free PMC article.
-
SDF-1/CXCR4 axis induces human dental pulp stem cell migration through FAK/PI3K/Akt and GSK3β/β-catenin pathways.Sci Rep. 2017 Jan 9;7:40161. doi: 10.1038/srep40161. Sci Rep. 2017. PMID: 28067275 Free PMC article.
-
Chemokine Regulation of Angiogenesis During Wound Healing.Adv Wound Care (New Rochelle). 2015 Nov 1;4(11):641-650. doi: 10.1089/wound.2014.0594. Adv Wound Care (New Rochelle). 2015. PMID: 26543678 Free PMC article. Review.
References
-
- Topol EJ. Current status and future prospects for acute myocardial infarction therapy. Circulation. 2003;108(Suppl 1):III6–III13. - PubMed
-
- Kawamoto A, et al. Intramyocardial transplantation of autologous endothelial progenitor cells for therapeutic neovascularization of myocardial ischemia. Circulation. 2003;107:461–468. - PubMed
-
- Gibson CM, et al. TIMI Study Group Relationship of the TIMI myocardial perfusion grades, flow grades, frame count, and percutaneous coronary intervention to long-term outcomes after thrombolytic administration in acute myocardial infarction. Circulation. 2002;105:1909–1913. - PubMed
-
- Wong GC, et al. Elevations in troponin T and I are associated with abnormal tissue level perfusion: A TACTICS-TIMI 18 substudy. Treat Angina with Aggrastat and Determine Cost of Therapy with an Invasive or Conservative Strategy-Thrombolysis in Myocardial Infarction. Circulation. 2002;106:202–207. - PubMed
-
- Yip HK, et al. Effect of the PercuSurge GuardWire device on the integrity of microvasculature and clinical outcomes during primary transradial coronary intervention in acute myocardial infarction. Am J Cardiol. 2003;92:1331–1335. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 HL077428/HL/NHLBI NIH HHS/United States
- R01 HL080137/HL/NHLBI NIH HHS/United States
- R01 HL057516/HL/NHLBI NIH HHS/United States
- HL-57516/HL/NHLBI NIH HHS/United States
- HL-53354/HL/NHLBI NIH HHS/United States
- HL-77428/HL/NHLBI NIH HHS/United States
- HL95874/HL/NHLBI NIH HHS/United States
- R01 HL095874/HL/NHLBI NIH HHS/United States
- R37 HL053354/HL/NHLBI NIH HHS/United States
- HL-63414/HL/NHLBI NIH HHS/United States
- R01 HL053354/HL/NHLBI NIH HHS/United States
- R01 HL063414/HL/NHLBI NIH HHS/United States
- HL-80137/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

