Cholecystokinin is up-regulated in obese mouse islets and expands beta-cell mass by increasing beta-cell survival
- PMID: 20534724
- PMCID: PMC2940525
- DOI: 10.1210/en.2010-0233
Cholecystokinin is up-regulated in obese mouse islets and expands beta-cell mass by increasing beta-cell survival
Abstract
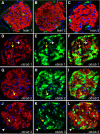
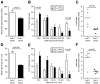
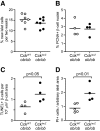
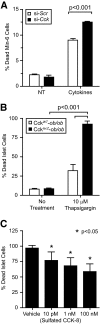
An absolute or functional deficit in beta-cell mass is a key factor in the pathogenesis of diabetes. We model obesity-driven beta-cell mass expansion by studying the diabetes-resistant C57BL/6-Leptin(ob/ob) mouse. We previously reported that cholecystokinin (Cck) was the most up-regulated gene in obese pancreatic islets. We now show that islet cholecystokinin (CCK) is up-regulated 500-fold by obesity and expressed in both alpha- and beta-cells. We bred a null Cck allele into the C57BL/6-Leptin(ob/ob) background and investigated beta-cell mass and metabolic parameters of Cck-deficient obese mice. Loss of CCK resulted in decreased islet size and reduced beta-cell mass through increased beta-cell death. CCK deficiency and decreased beta-cell mass exacerbated fasting hyperglycemia and reduced hyperinsulinemia. We further investigated whether CCK can directly affect beta-cell death in cell culture and isolated islets. CCK was able to directly reduce cytokine- and endoplasmic reticulum stress-induced cell death. In summary, CCK is up-regulated by islet cells during obesity and functions as a paracrine or autocrine factor to increase beta-cell survival and expand beta-cell mass to compensate for obesity-induced insulin resistance.
Figures

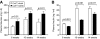
References
-
- Butler AE, Janson J, Bonner-Weir S, Ritzel R, Rizza RA, Butler PC 2003 β-Cell deficit and increased β-cell apoptosis in humans with type 2 diabetes. Diabetes 52:102–110 - PubMed
-
- Klöppel G, Löhr M, Habich K, Oberholzer M, Heitz PU 1985 Islet pathology and the pathogenesis of type 1 and type 2 diabetes mellitus revisited. Surv Synth Pathol Res 4:110–125 - PubMed
-
- Ritzel RA, Butler AE, Rizza RA, Veldhuis JD, Butler PC 2006 Relationship between β-cell mass and fasting blood glucose concentration in humans. Diabetes Care 29:717–718 - PubMed
-
- Keller MP, Choi Y, Wang P, Davis DB, Rabaglia ME, Oler AT, Stapleton DS, Argmann C, Schueler KL, Edwards S, Steinberg HA, Chaibub Neto E, Kleinhanz R, Turner S, Hellerstein MK, Schadt EE, Yandell BS, Kendziorski C, Attie AD 2008 A gene expression network model of type 2 diabetes links cell cycle regulation in islets with diabetes susceptibility. Genome Res 18:706–716 - PMC - PubMed
-
- Bock T, Pakkenberg B, Buschard K 2003 Increased islet volume but unchanged islet number in ob/ob mice. Diabetes 52:1716–1722 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

