A pilot study of volume measurement as a method of tumor response evaluation to aid biomarker development
- PMID: 20534736
- PMCID: PMC2940965
- DOI: 10.1158/1078-0432.CCR-10-0125
A pilot study of volume measurement as a method of tumor response evaluation to aid biomarker development
Abstract
Purpose: Tissue biomarker discovery is potentially limited by conventional tumor measurement techniques, which have an uncertain ability to accurately distinguish sensitive and resistant tumors. Semiautomated volumetric measurement of computed tomography imaging has the potential to more accurately capture tumor growth dynamics, allowing for more exact separation of sensitive and resistant tumors and a more accurate comparison of tissue characteristics.
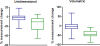
Experimental design: Forty-eight patients with early stage non-small cell lung cancer and clinical characteristics of sensitivity to gefitinib were studied. High-resolution computed tomography was done at baseline and after 3 weeks of gefitinib. Tumors were then resected and molecularly profiled. Unidimensional and volumetric measurements were done using a semiautomated algorithm. Measurement changes were evaluated for their ability to differentiate tumors with and without sensitizing mutations.
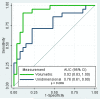
Results: Forty-four percent of tumors had epidermal growth factor receptor-sensitizing mutations. Receiver operating characteristic curve analysis showed that volumetric measurement had a higher area under the curve than unidimensional measurement for identifying tumors harboring sensitizing mutations (P = 0.009). Tumor volume decrease of >24.9% was the imaging criteria best able to classify tumors with and without sensitizing mutations (sensitivity, 90%; specificity, 89%).
Conclusions: Volumetric tumor measurement was better than unidimensional tumor measurement at distinguishing tumors based on presence or absence of a sensitizing mutation. Use of volume-based response assessment for the development of tissue biomarkers could reduce contamination between sensitive and resistant tumor populations, improving our ability to identify meaningful predictors of sensitivity.
Trial registration: ClinicalTrials.gov NCT00588445.
©2010 AACR.
Figures

Comment in
-
Volumes to learn: advancing therapeutics with innovative computed tomography image data analysis.Clin Cancer Res. 2010 Sep 15;16(18):4493-5. doi: 10.1158/1078-0432.CCR-10-1548. Epub 2010 Jul 19. Clin Cancer Res. 2010. PMID: 20643780 Free PMC article.
References
-
- Therasse P, Arbuck SG, Eisenhauer EA, et al. New guidelines to evaluate the response to treatment in solid tumors. Journal of National Cancer Institute. 2000;92:205–16. - PubMed
-
- Miller AB, Hoogstraten B, Staquet M, Winkler A. Reporting results of cancer treatment. Cancer. 1981;47:207–14. - PubMed
-
- Lara PN, Jr., Redman MW, Kelly K, et al. Disease control rate at 8 weeks predicts clinical benefit in advanced non-small-cell lung cancer: results from Southwest Oncology Group randomized trials. J Clin Oncol. 2008;26:463–7. - PubMed
-
- Birchard KR, Hoang JK, Herndon JE, Jr., Patz EF., Jr. Early changes in tumor size in patients treated for advanced stage nonsmall cell lung cancer do not correlate with survival. Cancer. 2009;115:581–6. - PubMed
-
- James K, Eisenhauer E, Christian M, et al. Measuring response in solid tumors: unidimensional versus bidimensional measurement. J Natl Cancer Inst. 1999;91:523–8. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

