Differential involvement of excitatory and inhibitory neurons of cat motor cortex in coincident spike activity related to behavioral context
- PMID: 20534853
- PMCID: PMC6632689
- DOI: 10.1523/JNEUROSCI.0770-10.2010
Differential involvement of excitatory and inhibitory neurons of cat motor cortex in coincident spike activity related to behavioral context
Abstract
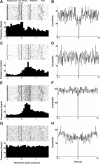
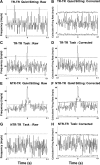
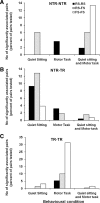
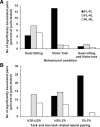
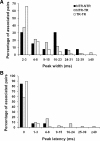
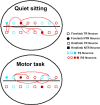
To assess temporal associations in spike activity between pairs of neurons in the primary motor cortex (MI) related to different behaviors, we compared the incidence of coincident spiking activity of task-related (TR) and non-task-related (NTR) neurons during a skilled motor task and sitting quietly in adult cats (Felis domestica). Chronically implanted microwires were used to record spike activity of MI neurons in four animals (two male and two female) trained to perform a skilled reaching task or sit quietly. Neurons were identified as TR if spike activity was modulated during the task (and NTR if not). Based on spike characteristics, they were also classified as either regular-spiking (RS, putatively excitatory) or fast-spiking (FS, putatively inhibitory) neurons. Temporal associations in the activities of simultaneously recorded neurons were evaluated using shuffle-corrected cross-correlograms. Pairs of NTR and TR neurons showed associations in their firing patterns over wide areas of MI (representing forelimb and hindlimb movements) during quiet sitting, more commonly involving RS neurons. During skilled task performance, however, significantly coincident firing was seen almost exclusively between TR neurons in a smaller part of MI (representing forelimb movements), involving mainly FS neurons. The findings of this study show evidence for widespread interactions in MI when the animal sits quietly, which changes to a more specific and restricted pattern of interactions during task performance. Different populations of excitatory and inhibitory neurons appear to be synchronized during skilled movement and quiet sitting.
Figures



References
-
- Allum JH, Hepp-Reymond M-C, Gysin R. Cross-correlation analysis of interneuronal connectivity in the motor cortex of the monkey. Brain Res. 1982;231:325–334. - PubMed
-
- Baker SN, Spinks R, Jackson A, Lemon RN. Synchronization in monkey motor cortex during a precision grip task. I. Task dependant modulation in single unit synchrony. J Neurophysiol. 2001;85:869–885. - PubMed
-
- Baranyi A, Szente MB, Woody CD. Electrophysiological characterization of different types of neurons recorded in vivo in the motor cortex of the cat. I. Patterns of firing activity and synaptic responses. J Neurophysiol. 1993;69:1850–1864. - PubMed
-
- Biswal B, Yetkin FZ, Haughton VM, Hyde JS. Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn Reson Med. 1995;34:537–541. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
