Conformational stability of Syrian hamster prion protein PrP(90-231)
- PMID: 20536231
- PMCID: PMC2902166
- DOI: 10.1021/ja100243h
Conformational stability of Syrian hamster prion protein PrP(90-231)
Abstract
Many transmissible spongiform encephalopathies (TSEs) are believed to be caused by a misfolded form of the normal cellular prion protein (PrP(C)) known as PrP(Sc). While PrP(Sc) is known to be exceptionally stable and resistant to protease degradation, PrP(C) has not shown these same unusual characteristics. However, using ion mobility spectrometry mass spectrometry (IMS-MS), we found evidence for at least one very stable conformation of a truncated form of recombinant PrP(C) consisting of residues 90-231, which resists unfolding in the absence of solvent at high injection energies and at temperatures in excess of 600 K. We also report the first absolute collision cross sections measured for recombinant Syrian hamster prion protein PrP(90-231).
Figures


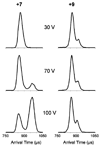

Similar articles
-
Structural analysis of prion proteins by means of drift cell and traveling wave ion mobility mass spectrometry.J Am Soc Mass Spectrom. 2010 May;21(5):845-54. doi: 10.1016/j.jasms.2010.01.017. Epub 2010 Jan 28. J Am Soc Mass Spectrom. 2010. PMID: 20206551
-
Molecular dynamics simulations of human prion protein: importance of correct treatment of electrostatic interactions.Biochemistry. 1999 Oct 19;38(42):13862-76. doi: 10.1021/bi991469d. Biochemistry. 1999. PMID: 10529232
-
Modulation of prion protein oligomerization, aggregation, and beta-sheet conversion by 4,4'-dianilino-1,1'-binaphthyl-5,5'-sulfonate (bis-ANS).J Biol Chem. 2004 Feb 13;279(7):5346-52. doi: 10.1074/jbc.M312262200. Epub 2003 Nov 21. J Biol Chem. 2004. PMID: 14634010
-
Prion protein oligomer and its neurotoxicity.Acta Biochim Biophys Sin (Shanghai). 2013 Jun;45(6):442-51. doi: 10.1093/abbs/gmt037. Epub 2013 Apr 4. Acta Biochim Biophys Sin (Shanghai). 2013. PMID: 23557632 Review.
-
Prion encephalopathies of animals and humans.Dev Biol Stand. 1993;80:31-44. Dev Biol Stand. 1993. PMID: 8270114 Review.
Cited by
-
Oligomerization of the microtubule-associated protein tau is mediated by its N-terminal sequences: implications for normal and pathological tau action.J Neurochem. 2016 Jun;137(6):939-54. doi: 10.1111/jnc.13604. Epub 2016 Apr 20. J Neurochem. 2016. PMID: 26953146 Free PMC article.
-
Ion mobility-mass spectrometry of a rotary ATPase reveals ATP-induced reduction in conformational flexibility.Nat Chem. 2014 Mar;6(3):208-215. doi: 10.1038/nchem.1868. Epub 2014 Feb 16. Nat Chem. 2014. PMID: 24557135 Free PMC article.
-
Aβ(39-42) modulates Aβ oligomerization but not fibril formation.Biochemistry. 2012 Jan 10;51(1):108-17. doi: 10.1021/bi201520b. Epub 2011 Dec 23. Biochemistry. 2012. PMID: 22129303 Free PMC article.
-
Ag+ Ion Binding to Human Metallothionein-2A Is Cooperative and Domain Specific.Anal Chem. 2020 Jul 7;92(13):8923-8932. doi: 10.1021/acs.analchem.0c00829. Epub 2020 Jun 24. Anal Chem. 2020. PMID: 32515580 Free PMC article.
-
High-Throughput Native Mass Spectrometry Screening in Drug Discovery.Front Mol Biosci. 2022 Apr 14;9:837901. doi: 10.3389/fmolb.2022.837901. eCollection 2022. Front Mol Biosci. 2022. PMID: 35495635 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

