Interactions between focused synaptic inputs and diffuse neuromodulation in the spinal cord
- PMID: 20536918
- PMCID: PMC3794674
- DOI: 10.1111/j.1749-6632.2010.05430.x
Interactions between focused synaptic inputs and diffuse neuromodulation in the spinal cord
Abstract
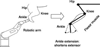
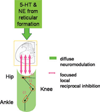
Spinal motoneurons (MNs) amplify synaptic inputs by producing strong dendritic persistent inward currents (PICs), which allow the MN to generate the firing rates and forces necessary for normal behaviors. However, PICs prolong MN depolarization after the initial excitation is removed, tend to "wind-up" with repeated activation and are regulated by a diffuse neuromodulatory system that affects all motor pools. We have shown that PICs are very sensitive to reciprocal inhibition from Ia afferents of antagonist muscles and as a result PIC amplification is related to limb configuration. Because reciprocal inhibition is tightly focused, shared only between strict anatomical antagonists, this system opposes the diffuse effects of the descending neuromodulation that facilitates PICs. Because inhibition appears necessary for PIC control, we hypothesize that Ia inhibition interacts with Ia excitation in a "push-pull" fashion, in which a baseline of simultaneous excitation and inhibition allows depolarization to occur via both excitation and disinhibition (and vice versa for hyperpolarization). Push-pull control appears to mitigate the undesirable affects associated with the PIC while still taking full advantage of PIC amplification.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures






Similar articles
-
Movement-related receptive fields of spinal motoneurones with active dendrites.J Physiol. 2008 Mar 15;586(6):1581-93. doi: 10.1113/jphysiol.2007.149146. Epub 2008 Jan 31. J Physiol. 2008. PMID: 18238818 Free PMC article.
-
Active properties of motoneurone dendrites: diffuse descending neuromodulation, focused local inhibition.J Physiol. 2008 Mar 1;586(5):1225-31. doi: 10.1113/jphysiol.2007.145078. Epub 2007 Oct 18. J Physiol. 2008. PMID: 17947305 Free PMC article. Review.
-
Synaptic integration in motoneurons with hyper-excitable dendrites.Can J Physiol Pharmacol. 2004 Aug-Sep;82(8-9):549-55. doi: 10.1139/y04-046. Can J Physiol Pharmacol. 2004. PMID: 15523512 Review.
-
Adjustable amplification of synaptic input in the dendrites of spinal motoneurons in vivo.J Neurosci. 2000 Sep 1;20(17):6734-40. doi: 10.1523/JNEUROSCI.20-17-06734.2000. J Neurosci. 2000. PMID: 10964980 Free PMC article.
-
Persistent inward currents in spinal motoneurons and their influence on human motoneuron firing patterns.Neuroscientist. 2008 Jun;14(3):264-75. doi: 10.1177/1073858408314986. Epub 2008 Apr 1. Neuroscientist. 2008. PMID: 18381974 Free PMC article. Review.
Cited by
-
Altered activation patterns by triceps surae stretch reflex pathways in acute and chronic spinal cord injury.J Neurophysiol. 2011 Oct;106(4):1669-78. doi: 10.1152/jn.00504.2011. Epub 2011 Jul 6. J Neurophysiol. 2011. PMID: 21734111 Free PMC article.
-
Frequency-dependent amplification of stretch-evoked excitatory input in spinal motoneurons.J Neurophysiol. 2012 Aug 1;108(3):753-9. doi: 10.1152/jn.00313.2012. Epub 2012 May 16. J Neurophysiol. 2012. PMID: 22592308 Free PMC article.
-
Differences in estimated persistent inward currents between ankle flexors and extensors in humans.J Neurophysiol. 2020 Aug 1;124(2):525-535. doi: 10.1152/jn.00746.2019. Epub 2020 Jul 15. J Neurophysiol. 2020. PMID: 32667263 Free PMC article.
-
Vibration attenuates spasm-like activity in humans with spinal cord injury.J Physiol. 2020 Jul;598(13):2703-2717. doi: 10.1113/JP279478. Epub 2020 Jun 10. J Physiol. 2020. PMID: 32298483 Free PMC article.
-
Voltage-dependent amplification of synaptic inputs in respiratory motoneurones.J Physiol. 2012 Jul 1;590(13):3067-90. doi: 10.1113/jphysiol.2011.225789. Epub 2012 Apr 10. J Physiol. 2012. PMID: 22495582 Free PMC article.
References
-
- Cushing S, Bui T, Rose PK. Effect of nonlinear summation of synaptic currents on the input-output properties of spinal motoneurons. J. Neurophysiol. 2005;94:3465–3478. - PubMed
-
- Sjostrom PJ, Rancz EA, Roth A, Hausser M. Dendritic excitability and synaptic plasticity. Physiol. Rev. 2008;88:769–840. - PubMed
-
- Baldissera F, Hultborn H, Illert M. Integration in spinal neuronal systems. In: MotorControl,part 1. V.B. Brooks, editor. Handbook of Physiology. Section 1 : The Nervous System. Vol. 2. Bethesda: American Physiological Society; 1981. pp. 509–595.
-
- Binder MD, Heckman CJ, Powers CK. Relative strengths and distributions of different sources pf synaptic input to the motoneurone pool: implications for motor unit recruitment. Adv. Exp. Med. Biol. 2002;508:207–212. - PubMed
-
- Heckman CJ, Lee RH, Brownstone RM. Hyperexcitable dendrites in motoneurons and their neuromodulatory control during motor behavior. Trends Neurosci. 2003;26:688–695. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

