Surrogate endpoints for overall survival in digestive oncology trials: which candidates? A questionnaires survey among clinicians and methodologists
- PMID: 20537166
- PMCID: PMC2904280
- DOI: 10.1186/1471-2407-10-277
Surrogate endpoints for overall survival in digestive oncology trials: which candidates? A questionnaires survey among clinicians and methodologists
Abstract
Background: Overall survival (OS) is the gold standard for the demonstration of a clinical benefit in cancer trials. Replacement of OS by a surrogate endpoint allows to reduce trial duration. To date, few surrogate endpoints have been validated in digestive oncology. The aim of this study was to draw up an ordered list of potential surrogate endpoints for OS in digestive cancer trials, by way of a survey among clinicians and methodologists. Secondary objective was to obtain their opinion on surrogacy and quality of life (QoL).
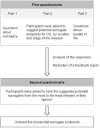
Methods: In 2007 and 2008, self administered sequential questionnaires were sent to a panel of French clinicians and methodologists involved in the conduct of cancer clinical trials. In the first questionnaire, panellists were asked to choose the most important characteristics defining a surrogate among six proposals, to give advantages and drawbacks of the surrogates, and to answer questions about their validation and use. Then they had to suggest potential surrogate endpoints for OS in each of the following tumour sites: oesophagus, stomach, liver, pancreas, biliary tract, lymphoma, colon, rectum, and anus. They finally gave their opinion on QoL as surrogate endpoint. In the second questionnaire, they had to classify the previously proposed candidate surrogates from the most (position #1) to the least relevant in their opinion.Frequency at which the endpoints were chosen as first, second or third most relevant surrogates was calculated and served as final ranking.
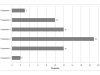
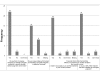
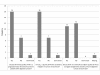
Results: Response rate was 30% (24/80) in the first round and 20% (16/80) in the second one. Participants highlighted key points concerning surrogacy. In particular, they reminded that a surrogate endpoint is expected to predict clinical benefit in a well-defined therapeutic situation. Half of them thought it was not relevant to study QoL as surrogate for OS.DFS, in the neoadjuvant settings or early stages, and PFS, in the non operable or metastatic settings, were ranked first, with a frequency of more than 69% in 20 out of 22 settings. PFS was proposed in association with QoL in metastatic primary liver and stomach cancers (both 81%). This composite endpoint was ranked second in metastatic oesophageal (69%), colorectal (56%) and anal (56%) cancers, whereas QoL alone was also suggested in most metastatic situations.Other endpoints frequently suggested were R0 resection in the neoadjuvant settings (oesophagus (69%), stomach (56%), pancreas (75%) and biliary tract (63%)) and response. An unexpected endpoint was metastatic PFS in non operable oesophageal (31%) and pancreatic (44%) cancers. Quality and results of surgical procedures like sphincter preservation were also cited as eligible surrogate endpoints in rectal (19%) and anal (50% in case of localized disease) cancers. Except for alpha-FP kinetic in hepatocellular carcinoma (13%) and CA19-9 decline (6%) in pancreas, few endpoints based on biological or tumour markers were proposed.
Conclusion: The overall results should help prioritise the endpoints to be statistically evaluated as surrogate for OS, so that trialists and clinicians can rely on endpoints that ensure relevant clinical benefit to the patient.
Figures
References
-
- Di Leo A, Buyse M, Bleiberg H. Is overall survival a realistic primary end point in advanced colorectal cancer studies? A critical assessment based on four clinical trials comparing fluorouracil plus leucovorin with the same treatment combined either with oxaliplatin or with CPT-11. Ann Oncol. 2004;15(4):545–549. doi: 10.1093/annonc/mdh127. - DOI - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

