Autoimmune-associated PTPN22 R620W variation reduces phosphorylation of lymphoid phosphatase on an inhibitory tyrosine residue
- PMID: 20538612
- PMCID: PMC2924087
- DOI: 10.1074/jbc.M110.111104
Autoimmune-associated PTPN22 R620W variation reduces phosphorylation of lymphoid phosphatase on an inhibitory tyrosine residue
Abstract
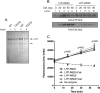
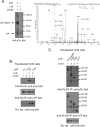
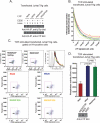
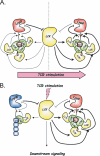
A missense C1858T single nucleotide polymorphism in the PTPN22 gene recently emerged as a major risk factor for human autoimmunity. PTPN22 encodes the lymphoid tyrosine phosphatase (LYP), which forms a complex with the kinase Csk and is a critical negative regulator of signaling through the T cell receptor. The C1858T single nucleotide polymorphism results in the LYP-R620W variation within the LYP-Csk interaction motif. LYP-W620 exhibits a greatly reduced interaction with Csk and is a gain-of-function inhibitor of signaling. Here we show that LYP constitutively interacts with its substrate Lck in a Csk-dependent manner. T cell receptor-induced phosphorylation of LYP by Lck on an inhibitory tyrosine residue releases tonic inhibition of signaling by LYP. The R620W variation disrupts the interaction between Lck and LYP, leading to reduced phosphorylation of LYP, which ultimately contributes to gain-of-function inhibition of T cell signaling.
Figures





References
-
- Bottini N., Musumeci L., Alonso A., Rahmouni S., Nika K., Rostamkhani M., MacMurray J., Meloni G. F., Lucarelli P., Pellecchia M., Eisenbarth G. S., Comings D., Mustelin T. (2004) Nat. Genet. 36, 337–338 - PubMed
-
- Begovich A. B., Carlton V. E., Honigberg L. A., Schrodi S. J., Chokkalingam A. P., Alexander H. C., Ardlie K. G., Huang Q., Smith A. M., Spoerke J. M., Conn M. T., Chang M., Chang S. Y., Saiki R. K., Catanese J. J., Leong D. U., Garcia V. E., McAllister L. B., Jeffery D. A., Lee A. T., Batliwalla F., Remmers E., Criswell L. A., Seldin M. F., Kastner D. L., Amos C. I., Sninsky J. J., Gregersen P. K. (2004) Am. J. Hum. Genet. 75, 330–337 - PMC - PubMed
-
- Bottini N., Vang T., Cucca F., Mustelin T. (2006) Semin. Immunol. 18, 207–213 - PubMed
-
- Gregersen P. K., Lee H. S., Batliwalla F., Begovich A. B. (2006) Semin. Immunol. 18, 214–223 - PubMed
-
- Todd J. A., Walker N. M., Cooper J. D., Smyth D. J., Downes K., Plagnol V., Bailey R., Nejentsev S., Field S. F., Payne F., Lowe C. E., Szeszko J. S., Hafler J. P., Zeitels L., Yang J. H., Vella A., Nutland S., Stevens H. E., Schuilenburg H., Coleman G., Maisuria M., Meadows W., Smink L. J., Healy B., Burren O. S., Lam A. A., Ovington N. R., Allen J., Adlem E., Leung H. T., Wallace C., Howson J. M., Guja C., Ionescu-Tîrgovişte C., Simmonds M. J., Heward J. M., Gough S. C., Dunger D. B., Wicker L. S., Clayton D. G. (2007) Nat. Genet. 39, 857–864 - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

