Involvement of auxin and brassinosteroid in the regulation of petiole elongation under the shade
- PMID: 20538889
- PMCID: PMC2923899
- DOI: 10.1104/pp.110.156802
Involvement of auxin and brassinosteroid in the regulation of petiole elongation under the shade
Abstract
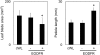
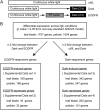
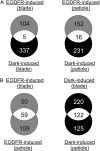
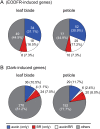
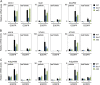
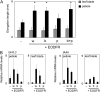
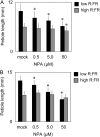
Plants grown under a canopy recognize changes in light quality and modify their growth patterns; this modification is known as shade avoidance syndrome. In leaves, leaf blade expansion is suppressed, whereas petiole elongation is promoted under the shade. However, the mechanisms that control these responses are largely unclear. Here, we demonstrate that both auxin and brassinosteroid (BR) are required for the normal leaf responses to shade in Arabidopsis (Arabidopsis thaliana). The microarray analysis of leaf blades and petioles treated with end-of-day far-red light (EODFR) revealed that almost half of the genes induced by the treatment in both parts were previously identified as auxin-responsive genes. Likewise, BR-responsive genes were overrepresented in the EODFR-induced genes. Hence, the auxin and BR responses were elevated by EODFR treatment in both leaf blades and petioles, although opposing growth responses were observed in these two parts. The analysis of the auxin-deficient doc1/big mutant and the BR-deficient rot3/cyp90c1 mutant further indicates that auxin and BR were equally required for the normal petiole elongation response to the shade stimulus. In addition, the spotlight irradiation experiment revealed that phytochrome in leaf blades but not that in petioles regulated petiole elongation, which was probably mediated through regulation of the auxin/BR responses in petioles. On the basis of these findings, we conclude that auxin and BR cooperatively promote petiole elongation in response to the shade stimulus under the control of phytochrome in the leaf blade.
Figures
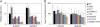
Similar articles
-
The different growth responses of the Arabidopsis thaliana leaf blade and the petiole during shade avoidance are regulated by photoreceptors and sugar.Plant Cell Physiol. 2005 Jan;46(1):213-23. doi: 10.1093/pcp/pci016. Epub 2005 Jan 19. Plant Cell Physiol. 2005. PMID: 15659441
-
ASYMMETRIC LEAVES1 promotes leaf hyponasty in Arabidopsis by light-mediated auxin signaling.Plant Physiol. 2024 Dec 23;197(1):kiae550. doi: 10.1093/plphys/kiae550. Plant Physiol. 2024. PMID: 39418078
-
Genetic control of petiole length in Arabidopsis thaliana.Plant Cell Physiol. 2002 Oct;43(10):1221-8. doi: 10.1093/pcp/pcf147. Plant Cell Physiol. 2002. PMID: 12407202
-
Seedling development in buckwheat and the discovery of the photomorphogenic shade-avoidance response.Plant Biol (Stuttg). 2013 Nov;15(6):931-40. doi: 10.1111/plb.12077. Epub 2013 Sep 24. Plant Biol (Stuttg). 2013. PMID: 24112603 Review.
-
Photomorphogenesis of leaves: shade-avoidance and differentiation of sun and shade leaves.Photochem Photobiol Sci. 2005 Sep;4(9):770-4. doi: 10.1039/b418440h. Epub 2005 May 4. Photochem Photobiol Sci. 2005. PMID: 16121290 Review.
Cited by
-
YUCCA auxin biosynthetic genes are required for Arabidopsis shade avoidance.PeerJ. 2016 Oct 13;4:e2574. doi: 10.7717/peerj.2574. eCollection 2016. PeerJ. 2016. PMID: 27761349 Free PMC article.
-
Organ-specific phytohormone synthesis in two Geranium species with antithetical responses to far-red light enrichment.Plant Direct. 2018 Aug 21;2(8):e00066. doi: 10.1002/pld3.66. eCollection 2018 Aug. Plant Direct. 2018. PMID: 31245741 Free PMC article.
-
Abscisic acid regulates axillary bud outgrowth responses to the ratio of red to far-red light.Plant Physiol. 2013 Oct;163(2):1047-58. doi: 10.1104/pp.113.221895. Epub 2013 Aug 8. Plant Physiol. 2013. PMID: 23929720 Free PMC article.
-
The Rice Basic Helix-Loop-Helix 79 (OsbHLH079) Determines Leaf Angle and Grain Shape.Int J Mol Sci. 2020 Mar 18;21(6):2090. doi: 10.3390/ijms21062090. Int J Mol Sci. 2020. PMID: 32197452 Free PMC article.
-
Stem transcriptome reveals mechanisms to reduce the energetic cost of shade-avoidance responses in tomato.Plant Physiol. 2012 Oct;160(2):1110-9. doi: 10.1104/pp.112.201921. Epub 2012 Aug 7. Plant Physiol. 2012. PMID: 22872775 Free PMC article.
References
-
- Asami T, Mizutani M, Fujioka S, Goda H, Min YK, Shimada Y, Nakano T, Takatsuto S, Matsuyama T, Nagata N, et al. (2001) Selective interaction of triazole derivatives with DWF4, a cytochrome P450 monooxygenase of the brassinosteroid biosynthetic pathway, correlates with brassinosteroid deficiency in planta. J Biol Chem 276: 25687–25691 - PubMed
-
- Benjamini Y, Hochberg Y. (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Statist Soc B 57: 289–300
-
- Black M, Shuttleworth JE. (1974) Role of cotyledons in photocontrol of hypocotyl extension in Cucumis sativus L. Planta 117: 57–66 - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

