Parstatin suppresses ocular neovascularization and inflammation
- PMID: 20538980
- PMCID: PMC3061514
- DOI: 10.1167/iovs.10-5576
Parstatin suppresses ocular neovascularization and inflammation
Abstract
Purpose: Parstatin is a 41-mer peptide formed by proteolytic cleavage on activation of the PAR1 receptor. The authors recently showed that parstatin is a potent inhibitor of angiogenesis. The purpose of the present study was to evaluate the therapeutic effect of parstatin on ocular neovascularization.
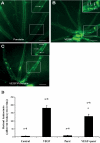
Methods: Choroidal neovascularization was generated in mice using laser-induced rupture of Bruch's membrane and was assessed after 14 days after perfusion of FITC-dextran. Oxygen-induced retinal neovascularization was established in neonatal mice by exposing them to 75% O(2) at postnatal day (P)7 for 5 days and then placing them in room air for 5 days. Evaluation was performed on P17 after staining with anti-mouse PECAM-1. The effect of parstatin was tested after intravitreal administration. The effects of subconjunctival-injected parstatin on corneal neovascularization and inflammation in rats were assessed 7 days after chemical burn-induced corneal neovascularization. Retinal leukostasis in mice was assessed after perfusion with FITC-conjugated concanavalin A.
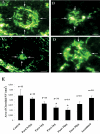
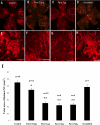
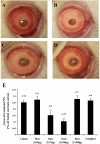
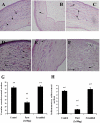
Results: Parstatin potently inhibited choroidal neovascularization with an IC(50) of approximately 3 μg and a maximum inhibition of 59% at 10 μg. Parstatin suppressed retinal neovascularization with maximum inhibition of 60% at 3 μg. Ten-microgram and 30-μg doses appeared to be toxic to the neonatal retina. Subconjunctival parstatin inhibited corneal neovascularization, with 200 μg the most effective dose (59% inhibition). In addition, parstatin significantly inhibited corneal inflammation and VEGF-induced retinal leukostasis. In all models tested, scrambled parstatin was without any significant effect.
Conclusions: Parstatin is a potent antiangiogenic agent of ocular neovascularization and may have clinical potential in the treatment of angiogenesis-related ocular disorders.
Figures
Similar articles
-
Effect of COX inhibitors on VEGF-induced retinal vascular leakage and experimental corneal and choroidal neovascularization.Exp Eye Res. 2004 Aug;79(2):275-85. doi: 10.1016/j.exer.2004.04.008. Exp Eye Res. 2004. PMID: 15325574
-
Influence of parstatin on experimental periodontal disease and repair in rats.J Periodontol. 2014 Sep;85(9):1266-74. doi: 10.1902/jop.2014.130619. Epub 2014 Jan 13. J Periodontol. 2014. PMID: 24410294
-
Apatinib, an Inhibitor of Vascular Endothelial Growth Factor Receptor 2, Suppresses Pathologic Ocular Neovascularization in Mice.Invest Ophthalmol Vis Sci. 2017 Jul 1;58(9):3592-3599. doi: 10.1167/iovs.17-21416. Invest Ophthalmol Vis Sci. 2017. PMID: 28715845
-
A potential therapeutic strategy for inhibition of ocular neovascularization with a new endogenous protein: rhEDI-8t.Graefes Arch Clin Exp Ophthalmol. 2012 May;250(5):731-9. doi: 10.1007/s00417-011-1765-y. Epub 2011 Sep 1. Graefes Arch Clin Exp Ophthalmol. 2012. PMID: 21881847
-
Animal models of ocular angiogenesis: from development to pathologies.FASEB J. 2017 Nov;31(11):4665-4681. doi: 10.1096/fj.201700336R. Epub 2017 Jul 24. FASEB J. 2017. PMID: 28739642 Free PMC article. Review.
Cited by
-
Anti-Parstatin Promotes Angiogenesis and Ameliorates Left Ventricular Dysfunction during Pressure Overload.Int J Biomed Sci. 2014 Mar;10(1):1-7. Int J Biomed Sci. 2014. PMID: 24711742 Free PMC article.
-
Blockade of VEGFR1 and 2 suppresses pathological angiogenesis and vascular leakage in the eye.PLoS One. 2011;6(6):e21411. doi: 10.1371/journal.pone.0021411. Epub 2011 Jun 22. PLoS One. 2011. PMID: 21731737 Free PMC article.
-
Current and emerging therapies for corneal neovascularization.Ocul Surf. 2018 Oct;16(4):398-414. doi: 10.1016/j.jtos.2018.06.004. Epub 2018 Jun 20. Ocul Surf. 2018. PMID: 29908870 Free PMC article. Review.
-
Signal Peptides and Their Fragments in Post-Translation: Novel Insights of Signal Peptides.Int J Mol Sci. 2024 Dec 18;25(24):13534. doi: 10.3390/ijms252413534. Int J Mol Sci. 2024. PMID: 39769297 Free PMC article. Review.
-
Calmodulin as a Key Regulator of Exosomal Signal Peptides.Cells. 2022 Dec 30;12(1):158. doi: 10.3390/cells12010158. Cells. 2022. PMID: 36611951 Free PMC article.
References
-
- Ferrara N, Kerbel RS. Angiogenesis as a therapeutic target. Nature. 2005;438:967–974 - PubMed
-
- Folkman J. Angiogenesis. Ann Rev Med. 2006;57:1–18 - PubMed
-
- Tolentino MJ. Current molecular understanding and future treatment strategies for pathologic ocular neovascularization. Curr Mol Med. 2009;9:973–981 - PubMed
-
- Coughlin SR. Protease-activated receptors in hemostasis, thrombosis, and vascular biology. J Thromb Haemost. 2005;3:1800–1814 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

