Metakaryotic stem cell lineages in organogenesis of humans and other metazoans
- PMID: 20539738
- PMCID: PMC2878747
- DOI: 10.4161/org.5.4.9632
Metakaryotic stem cell lineages in organogenesis of humans and other metazoans
Abstract
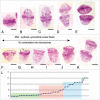
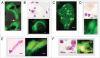
A non-eukaryotic, metakaryotic cell with large, open mouthed, bell shaped nuclei represents an important stem cell lineage in fetal/juvenile organogenesis in humans and rodents. each human bell shaped nucleus contains the diploid human DNA genome as tested by quantitative Feulgen DNA cytometry and fluorescent in situ hybridization with human pan-telomeric, pan-centromeric and chromosome specific probes. From weeks approximately 5-12 of human gestation the bell shaped nuclei are found in organ anlagen enclosed in sarcomeric tubular syncytia. Within syncytia bell shaped nuclear number increases binomially up to 16 or 32 nuclei; clusters of syncytia are regularly dispersed in organ anlagen. Syncytial bell shaped nuclei demonstrate two forms of symmetrical amitoses, facing or "kissing" bells and "stacking" bells resembling separation of two paper cups. Remarkably, DNA increase and nuclear fission occur coordinately. Importantly, syncytial bell shaped nuclei undergo asymmetrical amitoses creating organ specific ensembles of up to eight distinct closed nuclear forms, a characteristic required of a stem cell lineage. Closed nuclei emerging from bell shaped nuclei are eukaryotic as demonstrated by their subsequent increases by extra-syncytial mitoses populating the parenchyma of growing anlagen. From 9-14 weeks syncytia fragment forming single cells with bell shaped nuclei that continue to display both symmetrical and asymmetrical amitoses. These forms persist in the juvenile period and are specifically observed in bases of colonic crypts. Metakaryotic forms are found in organogenesis of humans, rats, mice and the plant Arabidopsis indicating an evolutionary origin prior to the divergence of plants and animals.
Keywords: human; metakaryote; metakaryotic cells; metazoans; organogenesis; stem cell lineage; stem cells.
Figures




References
-
- Gostjeva EV, Zukerberg L, Chung D, Thilly WG. Bell shaped nuclei dividing by symmetrical and asymmetrical nuclear fission have qualities of stem cells in human colonic embryogenesis and carcinogenesis. Cancer Genet Cytogenet. 2006;164:16–24. - PubMed
-
- Gostjeva EV, Thilly WG. Stem cell stages and the origins of colon cancer: a multi-disciplinary perspective. Stem Cell Rev. 2005;1:243–252. - PubMed
-
- Wilson EB. The Cell in Development and Inheritance. New York, MacMillan: 1896.
-
- Child CM. Amitosis as a factor in normal and regulatory growth. Anat Anz. 1907;30:271–297.
-
- Stough HB. Further studies of modified mitosis. J Morphol. 1935;58:221–256.
LinkOut - more resources
Full Text Sources
Other Literature Sources
