Smooth muscle differentiation and patterning in the urinary bladder
- PMID: 20541860
- PMCID: PMC3712869
- DOI: 10.1016/j.diff.2010.05.004
Smooth muscle differentiation and patterning in the urinary bladder
Abstract
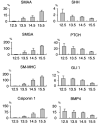
Smooth muscle differentiation and patterning is a fundamental process in urinary bladder development that involves a complex array of local environmental factors, epithelial-mesenchymal interaction, and signaling pathways. An epithelial signal is necessary to induce smooth muscle differentiation in the adjacent bladder mesenchyme. The bladder epithelium (urothelium) also influences the spatial organization of the bladder wall. Sonic hedgehog (Shh), which is expressed by the urothelium, promotes mesenchymal proliferation and induces differentiation of smooth muscle from embryonic bladder mesenchyme. Shh, whose signal is mediated through various transcription factors including Gli2 and BMP4, is likely also important in the patterning of bladder smooth muscle. However, it is not known to what extent early mediators of mesenchymal migration, other Shh-associated transcription factors, and crosstalk between the Shh signaling cascade and other pathways are involved in the patterning of bladder smooth muscle. Here we review the role of epithelial-mesenchymal interaction and Shh signaling in smooth muscle differentiation and patterning in the bladder. We also discuss emerging signaling molecules, transcription factors, and mesenchyme properties that might be fruitful areas of future research in the process of smooth muscle formation in the bladder.
Copyright © 2010 International Society of Differentiation. Published by Elsevier B.V. All rights reserved.
Figures







References
-
- Tanagho EA, Pugh RC. The anatomy and function of the ureterovesical junction. Br J Urol. 1963;35:151–165. - PubMed
-
- Tejedo-Mateu A, Vilanova-Trias J, Ruano-Gil D. Contribution to the study of the development of the terminal portion of the Wolffian duct and the ureter. Eur Urol. 1975;1:41–45. - PubMed
-
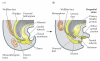
- Viana R, Batourina E, Huang H, Dressler GR, Kobayashi A, Behringer RR, Shapiro E, Hensle T, Lambert S, Mendelsohn C. The development of the bladder trigone, the center of the anti-reflux mechanism. Development. 2007;134:3763–3769. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

