The SUMO pathway functions in mouse oocyte maturation
- PMID: 20543581
- PMCID: PMC3322456
- DOI: 10.4161/cc.9.13.12120
The SUMO pathway functions in mouse oocyte maturation
Abstract
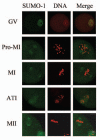
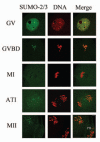
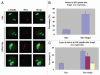
Sumoylation is an important post-translational modification in which SUMO (small ubiquitin-related modifier) proteins are bonded covalently to their substrates. Studies on the roles of sumoylation in cell cycle regulation have been emerging in both mitosis from yeast to mammals and meiosis in budding yeast, but the functions of sumoylation in mammalian meiosis, especially in oocyte meiotic maturation are not well known. Here, we examined the localization and expression of SUMO-1 and SUMO-2/3, the two basic proteins in the sumoylation pathway and investigated their roles through over-expression of Senp2 during mouse oocyte maturation. Immunofluorescent staining revealed differential patterns of SUMO-1 and SUMO-2/3 localization: SUMO-1 was localized to the spindle poles in prometaphase I, MI and MII stages, around the separating homologues in anaphase I and telophase I stages of first meiosis, while SUMO-2/3 was mainly concentrated near centromeres during mouse oocyte maturation. Immunoblot analysis uncovered the different expression profiles of SUMO-1 and SUMO-2/3 modified proteins during mouse oocyte maturation. Over-expression of Senp2, a SUMO-specific isopeptidase, caused changes of SUMO-modified proteins and led to defects in MII spindle organization in mature eggs. These results suggest that the SUMO pathway may play an indispensable role during mouse oocyte meiotic maturation.
© 2010 Landes Bioscience
Figures
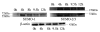

References
-
- Geiss-Friedlander R, Melchior F. Concepts in sumoylation: a decade on. Nat Rev Mol Cell Biol. 2007;8:947–956. - PubMed
-
- Meulmeester E, Melchior F. Cell biology: SUMO. Nature. 2008;452:709–711. - PubMed
-
- Mukhopadhyay D, Dasso M. Modification in reverse: the SUMO proteases. Trends Biochem Sci. 2007;32:286–295. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
