Astrocyte hypoxic response is essential for pathological but not developmental angiogenesis of the retina
- PMID: 20544853
- PMCID: PMC2993327
- DOI: 10.1002/glia.20997
Astrocyte hypoxic response is essential for pathological but not developmental angiogenesis of the retina
Abstract
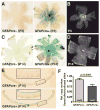
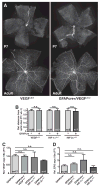
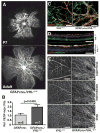
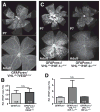
Vascular/parenchymal crosstalk is increasingly recognized as important in the development and maintenance of healthy vascularized tissues. The retina is an excellent model in which to study the role of cell type-specific contributions to the process of blood vessel and neuronal growth. During retinal vascular development, glial cells such as astrocytes provide the template over which endothelial cells migrate to form the retinal vascular network, and hypoxia-regulated vascular endothelial growth factor (VEGF) has been demonstrated to play a critical role in this process as well as pathological neovascularization. To investigate the nature of cell-specific contributions to this process, we deleted VEGF and its upstream regulators, the hypoxia-inducible transcription factors HIF-1 alpha and HIF-2 alpha, and the negative regulator of HIF alpha, von Hippel-Lindau protein (VHL), in astrocytes. We found that loss of hypoxic response and VEGF production in astrocytes does not impair normal development of retinal vasculature, indicating that astrocyte-derived VEGF is not essential for this process. In contrast, using a model of oxygen-induced ischemic retinopathy, we show that astrocyte-derived VEGF is essential for hypoxia-induced neovascularization. Thus, we demonstrate that astrocytes in the retina have highly divergent roles during developmental, physiological angiogenesis, and ischemia-driven, pathological neovascularization.
(c) 2010 Wiley-Liss, Inc.
Figures

Similar articles
-
Roles of HIFs and VEGF in angiogenesis in the retina and brain.J Clin Invest. 2019 Aug 12;129(9):3807-3820. doi: 10.1172/JCI126655. eCollection 2019 Aug 12. J Clin Invest. 2019. PMID: 31403471 Free PMC article.
-
The formation of an angiogenic astrocyte template is regulated by the neuroretina in a HIF-1-dependent manner.Dev Biol. 2012 Mar 1;363(1):106-14. doi: 10.1016/j.ydbio.2011.12.027. Epub 2011 Dec 24. Dev Biol. 2012. PMID: 22226979
-
Stabilization of myeloid-derived HIFs promotes vascular regeneration in retinal ischemia.Angiogenesis. 2020 May;23(2):83-90. doi: 10.1007/s10456-019-09681-1. Epub 2019 Oct 3. Angiogenesis. 2020. PMID: 31583505 Free PMC article.
-
Roles of Hypoxia Response in Retinal Development and Pathophysiology.Keio J Med. 2018 Mar 23;67(1):1-9. doi: 10.2302/kjm.2017-0002-IR. Epub 2017 Jun 6. Keio J Med. 2018. PMID: 28592747 Review.
-
Oxygen-dependent diseases in the retina: role of hypoxia-inducible factors.Exp Eye Res. 2006 Sep;83(3):473-83. doi: 10.1016/j.exer.2006.01.016. Epub 2006 Jun 5. Exp Eye Res. 2006. PMID: 16750526 Review.
Cited by
-
Hypoxia-induced angiogenesis: good and evil.Genes Cancer. 2011 Dec;2(12):1117-33. doi: 10.1177/1947601911423654. Genes Cancer. 2011. PMID: 22866203 Free PMC article.
-
Rank Order of Small Molecule Induced Hypoxiamimesis to Prevent Retinopathy of Prematurity.Front Cell Dev Biol. 2020 Jun 23;8:488. doi: 10.3389/fcell.2020.00488. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 32656210 Free PMC article.
-
Comparative Effects of Coenzyme Q10 or n-3 Polyunsaturated Fatty Acid Supplementation on Retinal Angiogenesis in a Rat Model of Oxygen-Induced Retinopathy.Antioxidants (Basel). 2018 Nov 9;7(11):160. doi: 10.3390/antiox7110160. Antioxidants (Basel). 2018. PMID: 30423931 Free PMC article.
-
Upregulation of retinal VEGF and connexin 43 in murine nonarteritic anterior ischemic optic neuropathy induced with 577 nm laser.Exp Eye Res. 2022 Dec;225:109139. doi: 10.1016/j.exer.2022.109139. Epub 2022 Jun 10. Exp Eye Res. 2022. PMID: 35691373 Free PMC article.
-
Comparative Analysis Reveals Novel Changes in Plasma Metabolites and Metabolomic Networks of Infants With Retinopathy of Prematurity.Invest Ophthalmol Vis Sci. 2022 Jan 3;63(1):28. doi: 10.1167/iovs.63.1.28. Invest Ophthalmol Vis Sci. 2022. PMID: 35060995 Free PMC article.
References
-
- Aiello LP, Pierce EA, Foley ED, Takagi H, Chen H, Riddle L, Ferrara N, King GL, Smith LE. Suppression of retinal neovascularization in vivo by inhibition of vascular endothelial growth factor (VEGF) using soluble VEGF-receptor chimeric proteins. Proc Natl Acad Sci U S A. 1995;92(23):10457–61. - PMC - PubMed
-
- Alon T, Hemo I, Itin A, Pe’er J, Stone J, Keshet E. Vascular endothelial growth factor acts as a survival factor for newly formed retinal vessels and has implications for retinopathy of prematurity. Nat Med. 1995;1(10):1024–8. - PubMed
-
- Arjamaa O, Nikinmaa M. Oxygen-dependent diseases in the retina: role of hypoxia-inducible factors. Exp Eye Res. 2006;83(3):473–83. - PubMed
-
- Banin E, Dorrell MI, Aguilar E, Ritter MR, Aderman CM, Smith AC, Friedlander J, Friedlander M. T2-TrpRS inhibits preretinal neovascularization and enhances physiological vascular regrowth in OIR as assessed by a new method of quantification. Invest Ophthalmol Vis Sci. 2006;47(5):2125–34. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

