Trypanosoma brucei pteridine reductase 1 is essential for survival in vitro and for virulence in mice
- PMID: 20545846
- PMCID: PMC2916222
- DOI: 10.1111/j.1365-2958.2010.07236.x
Trypanosoma brucei pteridine reductase 1 is essential for survival in vitro and for virulence in mice
Abstract
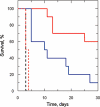
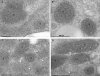
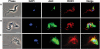
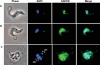
Gene knockout and knockdown methods were used to examine essentiality of pteridine reductase (PTR1) in pterin metabolism in the African trypanosome. Attempts to generate PTR1 null mutants in bloodstream form Trypanosoma brucei proved unsuccessful; despite integration of drug selectable markers at the target locus, the gene for PTR1 was either retained at the same locus or elsewhere in the genome. However, RNA interference (RNAi) resulted in complete knockdown of endogenous protein after 48 h, followed by cell death after 4 days. This lethal phenotype was reversed by expression of enzymatically active Leishmania major PTR1 in RNAi lines ((oe)RNAi) or by addition of tetrahydrobiopterin to cultures. Loss of PTR1 was associated with gross morphological changes due to a defect in cytokinesis, resulting in cells with multiple nuclei and kinetoplasts, as well as multiple detached flagella. Electron microscopy also revealed increased numbers of glycosomes, while immunofluorescence microscopy showed increased and more diffuse staining for glycosomal matrix enzymes, indicative of mis-localisation to the cytosol. Mis-localisation was confirmed by digitonin fractionation experiments. RNAi cell lines were markedly less virulent than wild-type parasites in mice and virulence was restored in the (oe)RNAi line. Thus, PTR1 may be a drug target for human African trypanosomiasis.
Figures




References
-
- Alibu VP, Storm L, Haile S, Clayton C, Horn D. A doubly inducible system for RNA interference and rapid RNAi plasmid construction in Trypanosoma brucei. Mol Biochem Parasitol. 2005;139:75–82. - PubMed
-
- Banerjee SK, Kessler PS, Saveria T, Parsons M. Identification of trypanosomatid PEX19: functional characterization reveals impact on cell growth and glycosome size and number. Mol Biochem Parasitol. 2005;142:47–55. - PubMed
-
- Berriman M, Ghedin E, Hertz-Fowler C, Blandin G, Renauld H, Bartholomeu DC, et al. The Genome of the African Trypanosome Trypanosoma brucei. Science. 2005;309:416–422. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

