Characterization of new ACADSB gene sequence mutations and clinical implications in patients with 2-methylbutyrylglycinuria identified by newborn screening
- PMID: 20547083
- PMCID: PMC2906669
- DOI: 10.1016/j.ymgme.2010.04.014
Characterization of new ACADSB gene sequence mutations and clinical implications in patients with 2-methylbutyrylglycinuria identified by newborn screening
Abstract
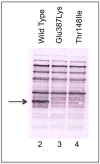
Short/branched chain acyl-CoA dehydrogenase (SBCAD) deficiency, also known as 2-methylbutyryl-CoA dehydrogenase deficiency, is a recently described autosomal recessive disorder of isoleucine metabolism. Most patients reported thus far have originated from a founder mutation in the Hmong Chinese population. While the first reported patients had severe disease, most of the affected Hmong have remained asymptomatic. In this study, we describe 11 asymptomatic non-Hmong patients brought to medical attention by elevated C5-carnitine found by newborn screening and one discovered because of clinical symptoms. The diagnosis of SBCAD deficiency was determined by metabolite analysis of blood, urine, and fibroblast samples. PCR and bidirectional sequencing were performed on genomic DNA from five of the patients covering the entire SBCAD (ACADSB) gene sequence of 11 exons. Sequence analysis of genomic DNA from each patient identified variations in the SBCAD gene not previously reported. Escherichia coli expression studies revealed that the missense mutations identified lead to inactivation or instability of the mutant SBCAD enzymes. These findings confirm that SBCAD deficiency can be identified through newborn screening by acylcarnitine analysis. Our patients have been well without treatment and call for careful follow-up studies to learn the true clinical impact of this disorder.
Copyright 2010 Elsevier Inc. All rights reserved.
Figures
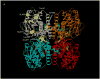
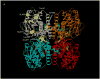
References
-
- Ikeda Y, Dabrowski C, Tanaka K. Separation and properties of five distinct acyl-CoA dehydrogenases from rat liver mitochondria. J Biol Chem. 1983;258:1066–1076. - PubMed
-
- Ikeda Y, Okamura-Ikeda K, Tanaka K. Purification and characterization of short-chain, medium-chain, and long-chain acyl-CoA dehydrogenases from rat liver mitochondria. Isolation of the holo- and apoenzymes and conversion of the apoenzyme to the holoenzyme. J Biol Chem. 1985;260:1311–1325. - PubMed
-
- Ikeda Y, Tanaka K. Purification and Characterization of 2-Methyl-Branched Chain Acyl Coenzyme A Dehydrogenase, an Enzyme Involved in Isoleucine and Valine Metabolism, from Rat Liver Mitochondria. J Biol Chem. 1983;258:9477–9487. - PubMed
-
- Izai K, Uchida Y, Orii T, Yamamoto S, Hashimoto T. Novel fatty acid β-oxidation enzymes in rat liver mitochondria. 1. Purification and properties of very-long-chain acyl-coenzyme A dehydrogenase. J Biol Chem. 1992;267:1027–1033. - PubMed
-
- Rozen R, Vockley J, Zhou L, Milos R, Willard J, Fu K, Vicanek C, Low-Nang L, Torban E, Fournier B. Isolation and expression of a cDNA encoding the precursor for a novel member (ACADSB) of the acyl-CoA dehydrogenase gene family. Genomics. 1994;24:280–287. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

