I-Ag7 is subject to post-translational chaperoning by CLIP
- PMID: 20547545
- PMCID: PMC2908477
- DOI: 10.1093/intimm/dxq056
I-Ag7 is subject to post-translational chaperoning by CLIP
Abstract
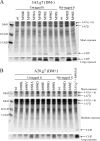
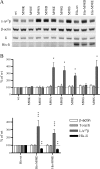
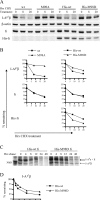
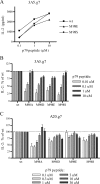
Several MHC class II alleles linked with autoimmune diseases form unusually low-stability complexes with class II-associated invariant chain peptides (CLIP), leading us to hypothesize that this is an important feature contributing to autoimmune pathogenesis. We recently demonstrated a novel post-endoplasmic reticulum (ER) chaperoning role of the CLIP peptides for the murine class II allele I-E(d). In the current study, we tested the generality of this CLIP chaperone function using a series of invariant chain (Ii) mutants designed to have varying CLIP affinity for I-A(g7). In cells expressing these Ii CLIP mutants, I-A(g7) abundance, turnover and antigen presentation are all subject to regulation by CLIP affinity, similar to I-E(d). However, I-A(g7) undergoes much greater quantitative changes than observed for I-E(d). In addition, we find that Ii with a CLIP region optimized for I-A(g7) binding may be preferentially assembled with I-A(g7) even in the presence of higher levels of wild-type Ii. This finding indicates that, although other regions of Ii interact with class II, CLIP binding to the groove is likely to be a dominant event in assembly of nascent class II molecules with Ii in the ER.
Figures
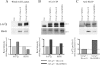
Similar articles
-
Posttranslational regulation of I-Ed by affinity for CLIP.J Immunol. 2007 Nov 1;179(9):5907-15. doi: 10.4049/jimmunol.179.9.5907. J Immunol. 2007. PMID: 17947664
-
Nonobese diabetic mice display elevated levels of class II-associated invariant chain peptide associated with I-Ag7 on the cell surface.J Immunol. 2001 Apr 1;166(7):4490-7. doi: 10.4049/jimmunol.166.7.4490. J Immunol. 2001. PMID: 11254705
-
Achieving stability through editing and chaperoning: regulation of MHC class II peptide binding and expression.Immunol Rev. 2005 Oct;207:242-60. doi: 10.1111/j.0105-2896.2005.00306.x. Immunol Rev. 2005. PMID: 16181341 Review.
-
T cell recognition of major histocompatibility complex class II complexes with invariant chain processing intermediates.J Exp Med. 1995 Nov 1;182(5):1403-13. doi: 10.1084/jem.182.5.1403. J Exp Med. 1995. PMID: 7595211 Free PMC article.
-
Ancient features of the MHC class II presentation pathway, and a model for the possible origin of MHC molecules.Immunogenetics. 2019 Mar;71(3):233-249. doi: 10.1007/s00251-018-1090-2. Epub 2018 Oct 30. Immunogenetics. 2019. PMID: 30377750 Review.
Cited by
-
MHC Class II Protein Turnover In vivo and Its Relevance for Autoimmunity in Non-Obese Diabetic Mice.Front Immunol. 2013 Nov 25;4:399. doi: 10.3389/fimmu.2013.00399. Front Immunol. 2013. PMID: 24324466 Free PMC article. Review.
-
Comparison of transduction efficiency among various lentiviruses containing GFP reporter in bone marrow hematopoietic stem cell transplantation.Exp Hematol. 2013 Nov;41(11):934-43. doi: 10.1016/j.exphem.2013.07.002. Epub 2013 Aug 14. Exp Hematol. 2013. PMID: 23954710 Free PMC article.
-
Transgene expression in various organs post BM-HSC transplantation.Stem Cell Res. 2014 Jan;12(1):209-21. doi: 10.1016/j.scr.2013.10.010. Epub 2013 Nov 2. Stem Cell Res. 2014. PMID: 24270160 Free PMC article.
-
Epitope Selection for HLA-DQ2 Presentation: Implications for Celiac Disease and Viral Defense.J Immunol. 2019 May 1;202(9):2558-2569. doi: 10.4049/jimmunol.1801454. Epub 2019 Mar 29. J Immunol. 2019. PMID: 30926644 Free PMC article.
-
On the perils of poor editing: regulation of peptide loading by HLA-DQ and H2-A molecules associated with celiac disease and type 1 diabetes.Expert Rev Mol Med. 2012 Jul 6;14:e15. doi: 10.1017/erm.2012.9. Expert Rev Mol Med. 2012. PMID: 22805744 Free PMC article. Review.
References
-
- Kouskoff V, Korganow AS, Duchatelle V, Degott C, Benoist C, Mathis D. Organ-specific disease provoked by systemic autoimmunity. Cell. 1996;87:811. - PubMed
-
- Kyburz D, Corr M. The KRN mouse model of inflammatory arthritis. Springer Semin. Immunopathol. 2003;25:79. - PubMed
-
- Liu GY, Baker D, Fairchild S, et al. Complete characterization of the expressed immune response genes in Biozzi AB/H mice: structural and functional identity between AB/H and NOD A region molecules. Immunogenetics. 1993;37:296. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

