Synergism between Wnt3a and heparin enhances osteogenesis via a phosphoinositide 3-kinase/Akt/RUNX2 pathway
- PMID: 20547765
- PMCID: PMC2924036
- DOI: 10.1074/jbc.M110.122069
Synergism between Wnt3a and heparin enhances osteogenesis via a phosphoinositide 3-kinase/Akt/RUNX2 pathway
Abstract
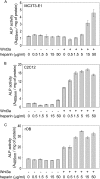
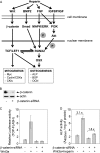
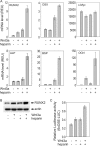
A new strategy has emerged to improve healing of bone defects using exogenous glycosaminoglycans by increasing the effectiveness of bone-anabolic growth factors. Wnt ligands play an important role in bone formation. However, their functional interactions with heparan sulfate/heparin have only been investigated in non-osseous tissues. Our study now shows that the osteogenic activity of Wnt3a is cooperatively stimulated through physical interactions with exogenous heparin. N-Sulfation and to a lesser extent O-sulfation of heparin contribute to the physical binding and optimal co-stimulation of Wnt3a. Wnt3a-heparin signaling synergistically increases osteoblast differentiation with minimal effects on cell proliferation. Thus, heparin selectively reduces the effective dose of Wnt3a needed to elicit osteogenic, but not mitogenic responses. Mechanistically, Wnt3a-heparin signaling strongly activates the phosphoinositide 3-kinase/Akt pathway and requires the bone-related transcription factor RUNX2 to stimulate alkaline phosphatase activity, which parallels canonical beta-catenin signaling. Collectively, our findings establish the osteo-inductive potential of a heparin-mediated Wnt3a-phosphoinositide 3-kinase/Akt-RUNX2 signaling network and suggest that heparan sulfate supplementation may selectively reduce the therapeutic doses of peptide factors required to promote bone formation.
Figures




References
-
- Guimond S., Maccarana M., Olwin B. B., Lindahl U., Rapraeger A. C. (1993) J. Biol. Chem. 268, 23906–23914 - PubMed
-
- Ishihara M., Shaklee P. N., Yang Z., Liang W., Wei Z., Stack R. J., Holme K. (1994) Glycobiology 4, 451–458 - PubMed
-
- Jackson R. A., Murali S., van Wijnen A. J., Stein G. S., Nurcombe V., Cool S. M. (2007) J. Cell. Physiol. 210, 38–50 - PubMed
-
- Lee J. Y., Choo J. E., Choi Y. S., Lee K. Y., Min D. S., Pi S. H., Seol Y. J., Lee S. J., Jo I. H., Chung C. P., Park Y. J. (2007) J. Biomed Mater. Res. A 83, 970–979 - PubMed
-
- Ling L., Murali S., Dombrowski C., Haupt L. M., Stein G. S., van Wijnen A. J., Nurcombe V., Cool S. M. (2006) J. Cell. Physiol. 209, 811–825 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

