Bile acids improve the antimicrobial effect of rifaximin
- PMID: 20547807
- PMCID: PMC2934964
- DOI: 10.1128/AAC.00161-10
Bile acids improve the antimicrobial effect of rifaximin
Abstract
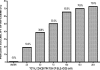
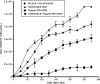
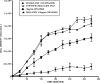
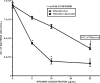
Diarrhea is one of the most common infirmities affecting international travelers, occurring in 20 to 50% of persons from industrialized countries visiting developing regions. Enterotoxigenic Escherichia coli (ETEC) is the most common causative agent and is isolated from approximately half of the cases of traveler's diarrhea. Rifaximin, a largely water-insoluble, nonabsorbable (<0.4%) antibiotic that inhibits bacterial RNA synthesis, is approved for use for the treatment of traveler's diarrhea caused by diarrheagenic E. coli. However, the drug has minimal effect on the bacterial flora or the infecting E. coli strain in the aqueous environment of the colon. The purpose of the present study was to evaluate the antimicrobial effect and bioavailability of rifaximin in aqueous solution in the presence and absence of physiologic concentrations of bile acids. The methods used included growth measurement of ETEC (strain H10407), rifaximin solubility measurements, total bacterial protein determination, and assessment of the functional activity of rifaximin by monitoring inhibition of bacterial beta-galactosidase expression. Solubility studies showed rifaximin to be 70- to 120-fold more soluble in bile acids (approximately 30% in 4 mM bile acids) than in aqueous solution. Addition of both purified bile acids and human bile to rifaximin at subinhibitory and inhibitory concentrations significantly improved the drug's anti-ETEC effect by 71% and 73%, respectively, after 4 h. This observation was confirmed by showing a decrease in the overall amount of total bacterial protein expressed during incubation of rifaximin plus bile acids. Rifaximin-treated samples containing bile acids inhibited the expression of ETEC beta-galactosidase at a higher magnitude than samples that did not contain bile acids. The study provides data showing that bile acids solubilize rifaximin on a dose-response basis, increasing the drug's bioavailability and antimicrobial effect. These observations suggest that rifaximin may be more effective in the treatment of infections in the small intestine, due to the higher concentration of bile in this region of the gastrointestinal tract than in the colon. The water insolubility of rifaximin is the likely explanation for the drug's minimal effects on colonic flora and fecal pathogens, despite in vitro susceptibility.
Figures

Similar articles
-
In vitro antimicrobial activity of rifaximin against enteropathogens causing traveler's diarrhea.Diagn Microbiol Infect Dis. 2007 Dec;59(4):473-5. doi: 10.1016/j.diagmicrobio.2007.07.003. Epub 2007 Sep 21. Diagn Microbiol Infect Dis. 2007. PMID: 17889485
-
Rifaximin: An Antibiotic with Important Biologic Effects.Mini Rev Med Chem. 2015;16(3):200-5. doi: 10.2174/1389557515666150722110201. Mini Rev Med Chem. 2015. PMID: 26202192 Review.
-
The potential for development of clinically relevant microbial resistance to rifaximin-α: a narrative review.Clin Microbiol Rev. 2023 Dec 20;36(4):e0003923. doi: 10.1128/cmr.00039-23. Epub 2023 Nov 16. Clin Microbiol Rev. 2023. PMID: 37971270 Free PMC article. Review.
-
In vitro antimicrobial susceptibility of bacterial enteropathogens isolated from international travelers to Mexico, Guatemala, and India from 2006 to 2008.Antimicrob Agents Chemother. 2011 Feb;55(2):874-8. doi: 10.1128/AAC.00739-10. Epub 2010 Nov 29. Antimicrob Agents Chemother. 2011. PMID: 21115800 Free PMC article.
-
Susceptibility of bacteria isolated from acute gastrointestinal infections to rifaximin and other antimicrobial agents in Mexico.Rev Gastroenterol Mex. 2016 Jan-Mar;81(1):3-10. doi: 10.1016/j.rgmx.2015.07.003. Epub 2015 Oct 30. Rev Gastroenterol Mex. 2016. PMID: 26525276 English, Spanish.
Cited by
-
Exploring molecular fingerprints of different drugs having bile interaction: a stepping stone towards better drug delivery.Mol Divers. 2024 Jun;28(3):1471-1483. doi: 10.1007/s11030-023-10670-2. Epub 2023 Jun 27. Mol Divers. 2024. PMID: 37369957
-
Eubiotic properties of rifaximin: Disruption of the traditional concepts in gut microbiota modulation.World J Gastroenterol. 2017 Jul 7;23(25):4491-4499. doi: 10.3748/wjg.v23.i25.4491. World J Gastroenterol. 2017. PMID: 28740337 Free PMC article. Review.
-
Microbial metabolite deoxycholic acid shapes microbiota against Campylobacter jejuni chicken colonization.PLoS One. 2019 Jul 5;14(7):e0214705. doi: 10.1371/journal.pone.0214705. eCollection 2019. PLoS One. 2019. PMID: 31276498 Free PMC article.
-
Gut Microbiota-Based Therapies for Irritable Bowel Syndrome.Clin Transl Gastroenterol. 2018 Feb 15;9(2):e134. doi: 10.1038/ctg.2018.2. Clin Transl Gastroenterol. 2018. PMID: 29446765 Free PMC article.
-
Novel therapeutic effects of rifaximin in combination with methylprednisolone for LPS-induced oxidative stress and inflammation in mice: An in vivo study.Toxicol Rep. 2024 Nov 12;13:101808. doi: 10.1016/j.toxrep.2024.101808. eCollection 2024 Dec. Toxicol Rep. 2024. PMID: 39640902 Free PMC article.
References
-
- Adachi, J. A., Z. D. Jiang, J. J. Mathewson, M. P. Verenkar, S. Thompson, F. Martinez-Sandoval, R. Steffen, C. D. Ericsson, and H. L. DuPont. 2001. Enteroaggregative Escherichia coli as a major etiologic agent in traveler's diarrhea in 3 regions of the world. Clin. Infect. Dis. 32:1706-1709. - PubMed
-
- Al-Abri, S. S., N. J. Beeching, and F. J. Nye. 2005. Traveler's diarrhoea. Lancet Infect. Dis. 5:349-360. - PubMed
-
- Begley, M., G. M. Cormac, and C. Hill. 2005. The interaction between bacteria and bile. FEMS Microbiol. Rev. 29:625-651. - PubMed
-
- Black, R. E. 1990. Epidemiology of travelers' diarrhea and relative importance of various pathogens. Rev. Infect. Dis. 12(Suppl. 1):S73-S79. - PubMed
-
- Black, R. E. 1993. Epidemiology of diarrhoeal disease: implications for control by vaccines. Vaccine 11:100-106. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

