Preferential localization of IgG memory B cells adjacent to contracted germinal centers
- PMID: 20547847
- PMCID: PMC2901464
- DOI: 10.1073/pnas.1005443107
Preferential localization of IgG memory B cells adjacent to contracted germinal centers
Abstract
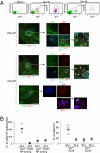
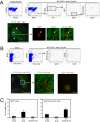
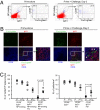
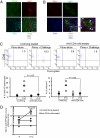
It has long been presumed that after leaving the germinal centers (GCs), memory B cells colonize the marginal zone or join the recirculating pool. Here we demonstrate the preferential localization of nitrophenol-chicken gamma-globulin-induced CD38(+)IgG1(+) memory B cells adjacent to contracted GCs in the spleen. The memory B cells in this region proliferated after secondary immunization, a response that was abolished by depletion of CD4(+) T cells. We also found that these IgG1(+) memory B cells could present antigen on their surface, and that this activity was required for their activation. These results implicate this peri-GC region as an important site for survival and reactivation of memory B cells.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Comment in
-
The persistence of memory: a unique niche for IgG memory B cells.Proc Natl Acad Sci U S A. 2010 Jul 20;107(29):12737-8. doi: 10.1073/pnas.1007717107. Epub 2010 Jul 12. Proc Natl Acad Sci U S A. 2010. PMID: 20624983 Free PMC article. No abstract available.
References
-
- Catron DM, Itano AA, Pape KA, Mueller DL, Jenkins MK. Visualizing the first 50 hr of the primary immune response to a soluble antigen. Immunity. 2004;21:341–347. - PubMed
-
- Garside P, et al. Visualization of specific B and T lymphocyte interactions in the lymph node. Science. 1998;281:96–99. - PubMed
-
- Allen CD, Okada T, Tang HL, Cyster JG. Imaging of germinal center selection events during affinity maturation. Science. 2007;315:528–531. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

