Polymorphic human prostaglandin H synthase-2 proteins and their interactions with cyclooxygenase substrates and inhibitors
- PMID: 20548327
- PMCID: PMC4465550
- DOI: 10.1038/tpj.2010.49
Polymorphic human prostaglandin H synthase-2 proteins and their interactions with cyclooxygenase substrates and inhibitors
Abstract
The cyclooxygenase (COX) activity of prostaglandin H synthase-2 (PGHS-2) is implicated in colorectal cancer and is targeted by nonsteroidal anti-inflammatory drugs (NSAIDs) and dietary n-3 fatty acids. We used purified, recombinant proteins to evaluate the functional impacts of the R228H, E488G, V511A and G587R PGHS-2 polymorphisms on COX activity, fatty acid selectivity and NSAID actions. Compared to wild-type PGHS-2, COX activity with arachidonate was ∼20% lower in 488G and ∼20% higher in 511A. All variants showed time-dependent inhibition by the COX-2-specific inhibitor (coxib) nimesulide, but 488G and 511A had 30-60% higher residual COX activity; 511A also showed up to 70% higher residual activity with other time-dependent inhibitors. In addition, 488G and 511A differed significantly from wild type in Vmax values with the two fatty acids: 488G showed ∼20% less and 511A showed ∼20% more discrimination against eicosapentaenoic acid. The Vmax value for eicosapentaenoate was not affected in 228H or 587R, nor were the Km values or the COX activation efficiency (with arachidonate) significantly altered in any variant. Thus, the E488G and V511A PGHS-2 polymorphisms may predict who will most likely benefit from interventions with some NSAIDs or n-3 fatty acids.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




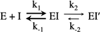
References
-
- Smith WL. Nutritionally essential fatty acids and biologically indispensable cyclooxygenases. Trends Biochem Sci. 2008;33:27–37. - PubMed
-
- Funk CD. Prostaglandins and leukotrienes: advances in eicosanoid biology. Science. 2001;294:1871–1875. - PubMed
-
- Simmons DL, Botting RM, Hla T. Cyclooxygenase isozymes: the biology of prostaglandin synthesis and inhibition. Pharmacol Rev. 2004;56:387–437. - PubMed
-
- Mann JR, DuBois RN. Cyclooxygenase-2 and gastrointestinal cancer. Cancer J. 2004;10:145–152. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

