Quercetin targets cysteine string protein (CSPalpha) and impairs synaptic transmission
- PMID: 20548785
- PMCID: PMC2883571
- DOI: 10.1371/journal.pone.0011045
Quercetin targets cysteine string protein (CSPalpha) and impairs synaptic transmission
Abstract
Background: Cysteine string protein (CSPalpha) is a synaptic vesicle protein that displays unique anti-neurodegenerative properties. CSPalpha is a member of the conserved J protein family, also called the Hsp40 (heat shock protein of 40 kDa) protein family, whose importance in protein folding has been recognized for many years. Deletion of the CSPalpha in mice results in knockout mice that are normal for the first 2-3 weeks of life followed by an unexplained presynaptic neurodegeneration and premature death. How CSPalpha prevents neurodegeneration is currently not known. As a neuroprotective synaptic vesicle protein, CSPalpha represents a promising therapeutic target for the prevention of neurodegenerative disorders.
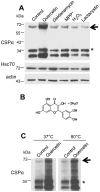
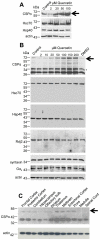
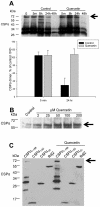
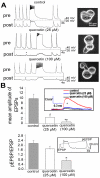
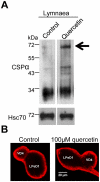
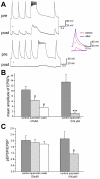
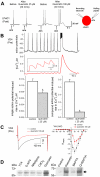
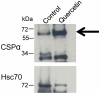
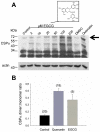
Methodology/principal findings: Here, we demonstrate that the flavonoid quercetin promotes formation of stable CSPalpha-CSPalpha dimers and that quercetin-induced dimerization is dependent on the unique cysteine string region. Furthermore, in primary cultures of Lymnaea neurons, quercetin induction of CSPalpha dimers correlates with an inhibition of synapse formation and synaptic transmission suggesting that quercetin interfers with CSPalpha function. Quercetin's action on CSPalpha is concentration dependent and does not promote dimerization of other synaptic proteins or other J protein family members and reduces the assembly of CSPalpha:Hsc70 units (70kDa heat shock cognate protein).
Conclusions/significance: Quercetin is a plant derived flavonoid and popular nutritional supplement proposed to prevent memory loss and altitude sickness among other ailments, although its precise mechanism(s) of action has been unclear. In view of the therapeutic promise of upregulation of CSPalpha and the undesired consequences of CSPalpha dysfunction, our data establish an essential proof of principle that pharmaceutical agents can selectively target the neuroprotective J protein CSPalpha.
Conflict of interest statement
Figures
Similar articles
-
CSPα knockout causes neurodegeneration by impairing SNAP-25 function.EMBO J. 2012 Feb 15;31(4):829-41. doi: 10.1038/emboj.2011.467. Epub 2011 Dec 20. EMBO J. 2012. PMID: 22187053 Free PMC article.
-
α-Synuclein A30P decreases neurodegeneration and increases synaptic vesicle release probability in CSPα-null mice.Neuropharmacology. 2014 Jan;76 Pt A:106-17. doi: 10.1016/j.neuropharm.2013.08.032. Epub 2013 Sep 11. Neuropharmacology. 2014. PMID: 24036317
-
CSPα promotes SNARE-complex assembly by chaperoning SNAP-25 during synaptic activity.Nat Cell Biol. 2011 Jan;13(1):30-9. doi: 10.1038/ncb2131. Epub 2010 Dec 12. Nat Cell Biol. 2011. PMID: 21151134
-
CSPalpha: the neuroprotective J protein.Biochem Cell Biol. 2010 Apr;88(2):157-65. doi: 10.1139/o09-124. Biochem Cell Biol. 2010. PMID: 20453918 Review.
-
Cysteine string protein (CSP) and its role in preventing neurodegeneration.Semin Cell Dev Biol. 2015 Apr;40:153-9. doi: 10.1016/j.semcdb.2015.03.008. Epub 2015 Mar 21. Semin Cell Dev Biol. 2015. PMID: 25800794 Free PMC article. Review.
Cited by
-
Exome-sequencing confirms DNAJC5 mutations as cause of adult neuronal ceroid-lipofuscinosis.PLoS One. 2011;6(11):e26741. doi: 10.1371/journal.pone.0026741. Epub 2011 Nov 4. PLoS One. 2011. PMID: 22073189 Free PMC article.
-
Cysteine string protein α and a link between rare and common neurodegenerative dementias.NPJ Dement. 2025;1(1):15. doi: 10.1038/s44400-025-00016-0. Epub 2025 Jul 3. NPJ Dement. 2025. PMID: 40621104 Free PMC article. Review.
-
Increased Expression of the Large Conductance, Calcium-Activated K+ (BK) Channel in Adult-Onset Neuronal Ceroid Lipofuscinosis.PLoS One. 2015 Apr 23;10(4):e0125205. doi: 10.1371/journal.pone.0125205. eCollection 2015. PLoS One. 2015. PMID: 25905915 Free PMC article.
-
Biological Effect of Quercetin in Repairing Brain Damage and Cerebral Changes in Rats: Molecular Docking and In Vivo Studies.Biomed Res Int. 2022 Apr 26;2022:8962149. doi: 10.1155/2022/8962149. eCollection 2022. Biomed Res Int. 2022. PMID: 35528172 Free PMC article.
-
The Plasmodium falciparum exported J domain proteins fine-tune human and malarial Hsp70s: pathological exploitation of proteostasis machinery.Front Mol Biosci. 2023 Jun 30;10:1216192. doi: 10.3389/fmolb.2023.1216192. eCollection 2023. Front Mol Biosci. 2023. PMID: 37457831 Free PMC article. Review.
References
-
- Mastrogiacomo A, Parsons SM, Zampighi GA, Jenden DJ, Umbach JA, et al. Cysteine string proteins: a potential link between synaptic vesicles and presynaptic Ca2+ channels. Science. 1994;263:981–982. - PubMed
-
- Zinsmaier KE, Eberle KK, Buchner E, Walter N, Benzer S. Paralysis and early death in cysteine string protein mutants of Drosophila. Science. 1994;263:977–980. - PubMed
-
- Fernandez-Chacon R, Wolfel M, Nishimune H, Tabares L, Schmitz F, et al. The synaptic vesicle protein CSP alpha prevents presynaptic degeneration. Neuron. 2004;42:237–251. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

