Cushing's syndrome and fetal features resurgence in adrenal cortex-specific Prkar1a knockout mice
- PMID: 20548949
- PMCID: PMC2883593
- DOI: 10.1371/journal.pgen.1000980
Cushing's syndrome and fetal features resurgence in adrenal cortex-specific Prkar1a knockout mice
Abstract
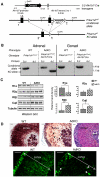
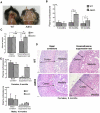
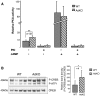
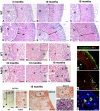
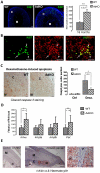
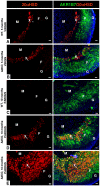
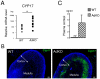
Carney complex (CNC) is an inherited neoplasia syndrome with endocrine overactivity. Its most frequent endocrine manifestation is primary pigmented nodular adrenocortical disease (PPNAD), a bilateral adrenocortical hyperplasia causing pituitary-independent Cushing's syndrome. Inactivating mutations in PRKAR1A, a gene encoding the type 1 alpha-regulatory subunit (R1alpha) of the cAMP-dependent protein kinase (PKA) have been found in 80% of CNC patients with Cushing's syndrome. To demonstrate the implication of R1alpha loss in the initiation and development of PPNAD, we generated mice lacking Prkar1a specifically in the adrenal cortex (AdKO). AdKO mice develop pituitary-independent Cushing's syndrome with increased PKA activity. This leads to autonomous steroidogenic genes expression and deregulated adreno-cortical cells differentiation, increased proliferation and resistance to apoptosis. Unexpectedly, R1alpha loss results in improper maintenance and centrifugal expansion of cortisol-producing fetal adrenocortical cells with concomitant regression of adult cortex. Our data provide the first in vivo evidence that loss of R1alpha is sufficient to induce autonomous adrenal hyper-activity and bilateral hyperplasia, both observed in human PPNAD. Furthermore, this model demonstrates that deregulated PKA activity favors the emergence of a new cell population potentially arising from the fetal adrenal, giving new insight into the mechanisms leading to PPNAD.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

Similar articles
-
PRKAR1A mutations in primary pigmented nodular adrenocortical disease.Pituitary. 2006;9(3):211-9. doi: 10.1007/s11102-006-0266-1. Pituitary. 2006. PMID: 17036196 Review.
-
Celecoxib reduces glucocorticoids in vitro and in a mouse model with adrenocortical hyperplasia.Endocr Relat Cancer. 2016 Jan;23(1):15-25. doi: 10.1530/ERC-15-0472. Epub 2015 Oct 5. Endocr Relat Cancer. 2016. PMID: 26438728 Free PMC article.
-
Mutations of the PRKAR1A gene in Cushing's syndrome due to sporadic primary pigmented nodular adrenocortical disease.J Clin Endocrinol Metab. 2002 Sep;87(9):4324-9. doi: 10.1210/jc.2002-020592. J Clin Endocrinol Metab. 2002. PMID: 12213893
-
Bilateral Adrenocortical Nodular Disease and Cushing's Syndrome.J Clin Endocrinol Metab. 2024 Sep 16;109(10):2422-2432. doi: 10.1210/clinem/dgae419. J Clin Endocrinol Metab. 2024. PMID: 38888184 Review.
-
Adrenal pathophysiology: lessons from the Carney complex.Horm Res. 2005;64(3):132-9. doi: 10.1159/000088586. Epub 2005 Sep 27. Horm Res. 2005. PMID: 16192737 Review.
Cited by
-
PKA inhibits WNT signalling in adrenal cortex zonation and prevents malignant tumour development.Nat Commun. 2016 Sep 14;7:12751. doi: 10.1038/ncomms12751. Nat Commun. 2016. PMID: 27624192 Free PMC article.
-
Mouse Models Recapitulating Human Adrenocortical Tumors: What Is Lacking?Front Endocrinol (Lausanne). 2016 Jul 15;7:93. doi: 10.3389/fendo.2016.00093. eCollection 2016. Front Endocrinol (Lausanne). 2016. PMID: 27471492 Free PMC article. Review.
-
A Novel Population of Inner Cortical Cells in the Adrenal Gland That Displays Sexually Dimorphic Expression of Thyroid Hormone Receptor-β1.Endocrinology. 2015 Jun;156(6):2338-48. doi: 10.1210/en.2015-1118. Epub 2015 Mar 16. Endocrinology. 2015. PMID: 25774556 Free PMC article.
-
Conditional mutagenesis of Gata6 in SF1-positive cells causes gonadal-like differentiation in the adrenal cortex of mice.Endocrinology. 2013 May;154(5):1754-67. doi: 10.1210/en.2012-1892. Epub 2013 Mar 7. Endocrinology. 2013. PMID: 23471215 Free PMC article.
-
Transgenic Mouse Models to Study the Development and Maintenance of the Adrenal Cortex.Int J Mol Sci. 2022 Nov 19;23(22):14388. doi: 10.3390/ijms232214388. Int J Mol Sci. 2022. PMID: 36430866 Free PMC article. Review.
References
-
- Carney JA, Gordon H, Carpenter PC, Shenoy BV, Go VL. The complex of myxomas, spotty pigmentation, and endocrine overactivity. Medicine (Baltimore) 1985;64:270–283. - PubMed
-
- Kirschner LS, Carney JA, Pack SD, Taymans SE, Giatzakis C, et al. Mutations of the gene encoding the protein kinase A type I-alpha regulatory subunit in patients with the Carney complex. Nat Genet. 2000;26:89–92. - PubMed
-
- Groussin L, Jullian E, Perlemoine K, Louvel A, Leheup B, et al. Mutations of the PRKAR1A gene in Cushing's syndrome due to sporadic primary pigmented nodular adrenocortical disease. J Clin Endocrinol Metab. 2002;87:4324–4329. - PubMed
-
- Amieux PS, Howe DG, Knickerbocker H, Lee DC, Su T, et al. Increased basal cAMP-dependent protein kinase activity inhibits the formation of mesoderm-derived structures in the developing mouse embryo. J Biol Chem. 2002;277:27294–27304. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

