Tumour-associated carbohydrate antigens in breast cancer
- PMID: 20550729
- PMCID: PMC2917018
- DOI: 10.1186/bcr2577
Tumour-associated carbohydrate antigens in breast cancer
Abstract
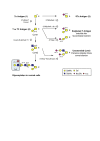
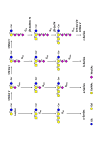
Glycosylation changes that occur in cancer often lead to the expression of tumour-associated carbohydrate antigens. In breast cancer, these antigens are usually associated with a poor prognosis and a reduced overall survival. Cellular models have shown the implication of these antigens in cell adhesion, migration, proliferation and tumour growth. The present review summarizes our current knowledge of glycosylation changes (structures, biosynthesis and occurrence) in breast cancer cell lines and primary tumours, and the consequences on disease progression and aggressiveness. The therapeutic strategies attempted to target tumour-associated carbohydrate antigens in breast cancer are also discussed.
Figures


References
-
- Le Marer N, Laudet V, Svensson EC, Cazlaris H, Van Hille B, Lagrou C, Stehelin D, Montreuil J, Verbert A, Delannoy P. The c-Ha-ras oncogene induces increased expression of beta-galactoside alpha-2, 6-sialyltransferase in rat fibroblast (FR3T3) cells. Glycobiology. 1992;2:49–56. doi: 10.1093/glycob/2.1.49. - DOI - PubMed
-
- Soares R, Marinho A, Schmitt F. Expression of sialyl-Tn in breast cancer. Correlation with prognostic parameters. Pathol Res Pract. 1996;192:1181–1186. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

