Knockdown of zebrafish lumican gene (zlum) causes scleral thinning and increased size of scleral coats
- PMID: 20551313
- PMCID: PMC2934679
- DOI: 10.1074/jbc.M109.043679
Knockdown of zebrafish lumican gene (zlum) causes scleral thinning and increased size of scleral coats
Abstract
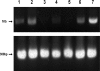
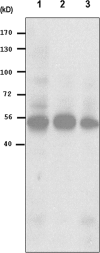
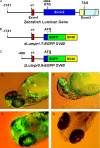
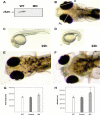
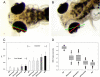
The lumican gene (lum), which encodes one of the major keratan sulfate proteoglycans (KSPGs) in the vertebrate cornea and sclera, has been linked to axial myopia in humans. In this study, we chose zebrafish (Danio rerio) as an animal model to elucidate the role of lumican in the development of axial myopia. The zebrafish lumican gene (zlum) spans approximately 4.6 kb of the zebrafish genome. Like human (hLUM) and mouse (mlum), zlum consists of three exons, two introns, and a TATA box-less promoter at the 5'-flanking region of the transcription initiation site. Sequence analysis of the cDNA predicts that zLum encodes 344 amino acids. zLum shares 51% amino acid sequence identity with human lumican. Similar to hLUM and mlum, zlum mRNA is expressed in the eye and many other tissues, such as brain, muscle, and liver as well. Transgenic zebrafish harboring an enhanced GFP reporter gene construct downstream of a 1.7-kb zlum 5'-flanking region displayed enhanced GFP expression in the cornea and sclera, as well as throughout the body. Down-regulation of zlum expression by antisense zlum morpholinos manifested ocular enlargement resembling axial myopia due to disruption of the collagen fibril arrangement in the sclera and resulted in scleral thinning. Administration of muscarinic receptor antagonists, e.g. atropine and pirenzepine, effectively subdued the ocular enlargement caused by morpholinos in in vivo zebrafish larvae assays. The observation suggests that zebrafish can be used as an in vivo model for screening compounds in treating myopia.
Figures






References
-
- Lin L. L., Shih Y. F., Hsiao C. K., Chen C. J., Lee L. A., Hung P. T. (2001) J. Formos. Med. Assoc. 100, 684–691 - PubMed
-
- Kempen J. H., Mitchell P., Lee K. E., Tielsch J. M., Broman A. T., Taylor H. R., Ikram M. K., Congdon N. G., O'Colmain B. J. (2004) Arch. Ophthalmol. 122, 495–505 - PubMed
-
- McBrien N. A., Metlapally R., Jobling A. I., Gentle A. (2006) Invest. Ophthalmol. Vis. Sci. 47, 4674–4682 - PubMed
-
- Deere M., Johnson J., Garza S., Harrison W. R., Yoon S. J., Elder F. F., Kucherlapati R., Hook M., Hecht J. T. (1996) Genomics 38, 399–404 - PubMed
-
- Fisher L. W., Heegaard A. M., Vetter U., Vogel W., Just W., Termine J. D., Young M. F. (1991) J. Biol. Chem. 266, 14371–14377 - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

