Structure, dynamics, and substrate-induced conformational changes of the multidrug transporter EmrE in liposomes
- PMID: 20551331
- PMCID: PMC2924113
- DOI: 10.1074/jbc.M110.132621
Structure, dynamics, and substrate-induced conformational changes of the multidrug transporter EmrE in liposomes
Abstract
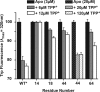
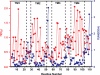
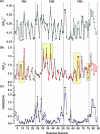
EmrE, a member of the small multidrug transporters superfamily, extrudes positively charged hydrophobic compounds out of Escherichia coli cytoplasm in exchange for inward movement of protons down their electrochemical gradient. Although its transport mechanism has been thoroughly characterized, the structural basis of energy coupling and the conformational cycle mediating transport have yet to be elucidated. In this study, EmrE structure in liposomes and the substrate-induced conformational changes were investigated by systematic spin labeling and EPR analysis. Spin label mobilities and accessibilities describe a highly dynamic ligand-free (apo) conformation. Dipolar coupling between spin labels across the dimer reveals at least two spin label populations arising from different packing interfaces of the EmrE dimer. One population is consistent with antiparallel arrangement of the monomers, although the EPR parameters suggest deviations from the crystal structure of substrate-bound EmrE. Resolving these discrepancies requires an unusual disposition of TM3 relative to the membrane-water interface and a kink in its backbone that enables bending of its C-terminal part. Binding of the substrate tetraphenylphosphonium changes the environment of spin labels and their proximity in three transmembrane helices. The underlying conformational transition involves repacking of TM1, tilting of TM2, and changes in the backbone configurations of TM3 and the adjacent loop connecting it to TM4. A dynamic apo conformation is necessary for the polyspecificity of EmrE allowing the binding of structurally diverse substrates. The flexibility of TM3 may play a critical role in movement of substrates across the membrane.
Figures





References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

