Calcineurin-mediated dephosphorylation of synaptotagmin VI is necessary for acrosomal exocytosis
- PMID: 20551332
- PMCID: PMC2924044
- DOI: 10.1074/jbc.M109.095752
Calcineurin-mediated dephosphorylation of synaptotagmin VI is necessary for acrosomal exocytosis
Abstract
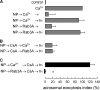
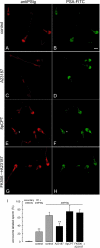
Regulated secretion is a fundamental process underlying the function of many cell types. In particular, acrosomal exocytosis in mammalian sperm is essential for egg fertilization. In general, exocytosis is initiated by a cytosolic calcium increase. In this report we show that calcium affects several factors during human sperm acrosomal exocytosis. By using an antibody that specifically recognizes synaptotagmin VI phosphorylated at the polybasic region of the C2B domain, we showed that a calcium-dependent dephosphorylation of this protein occurred at early stages of the acrosomal exocytosis in streptolysin O-permeabilized sperm. We identified the phosphatase as calcineurin and showed that the activity of this enzyme is absolutely required during the early steps of the secretory process. When added to sperm, an inhibitor-insensitive, catalytically active domain of calcineurin was able to rescue the effect of the specific calcineurin inhibitor cyclosporin A. This same domain dephosphorylated recombinant synaptotagmin VI C2B domain, validating this protein as a new substrate for calcineurin. When sperm were treated with catalytically active calcineurin before stimulation, exocytosis was inhibited, an effect that was rescued by the phosphomimetic synaptotagmin VI C2B-T418E,T419E mutant domain. These observations indicate that synaptotagmin must be dephosphorylated at a specific window of time and suggest that phosphorylated synaptotagmin has an active role at early stages of the acrosomal exocytosis.
Figures






Similar articles
-
Protein kinase C-mediated phosphorylation of the two polybasic regions of synaptotagmin VI regulates their function in acrosomal exocytosis.Dev Biol. 2005 Sep 15;285(2):422-35. doi: 10.1016/j.ydbio.2005.07.007. Dev Biol. 2005. PMID: 16111671
-
Complexin/synaptotagmin interplay controls acrosomal exocytosis.J Biol Chem. 2007 Sep 7;282(36):26335-43. doi: 10.1074/jbc.M700854200. Epub 2007 Jul 4. J Biol Chem. 2007. PMID: 17613520
-
Synaptotagmin VI participates in the acrosome reaction of human spermatozoa.Dev Biol. 2001 Jul 15;235(2):521-9. doi: 10.1006/dbio.2001.0316. Dev Biol. 2001. PMID: 11437455
-
The role of the acrosomal matrix in fertilization.Int J Dev Biol. 2008;52(5-6):511-22. doi: 10.1387/ijdb.072532mb. Int J Dev Biol. 2008. PMID: 18649264 Review.
-
Acrosomal exocytosis, a special type of regulated secretion.IUBMB Life. 2007 Apr-May;59(4-5):286-92. doi: 10.1080/15216540701222872. IUBMB Life. 2007. PMID: 17505967 Review.
Cited by
-
Isolation of a calcium-binding protein of the acrosomal membrane of bovine spermatozoa.Int J Biochem Cell Biol. 2013 Apr;45(4):876-84. doi: 10.1016/j.biocel.2013.01.016. Epub 2013 Jan 29. Int J Biochem Cell Biol. 2013. PMID: 23376657 Free PMC article.
-
PP1, PP2A and PP2B Interplay in the Regulation of Sperm Motility: Lessons from Protein Phosphatase Inhibitors.Int J Mol Sci. 2022 Dec 3;23(23):15235. doi: 10.3390/ijms232315235. Int J Mol Sci. 2022. PMID: 36499559 Free PMC article. Review.
-
Redistribution of soluble N-ethylmaleimide-sensitive-factor attachment protein receptors in mouse sperm membranes prior to the acrosome reaction.Biol Reprod. 2017 Feb 1;96(2):352-365. doi: 10.1095/biolreprod.116.143735. Biol Reprod. 2017. PMID: 28203732 Free PMC article.
-
Signaling Enzymes Required for Sperm Maturation and Fertilization in Mammals.Front Cell Dev Biol. 2019 Dec 18;7:341. doi: 10.3389/fcell.2019.00341. eCollection 2019. Front Cell Dev Biol. 2019. PMID: 31921853 Free PMC article. Review.
-
Calcineurin Aα Contributes to IgE-Dependent Mast-Cell Mediator Secretion in Allergic Inflammation.J Innate Immun. 2022;14(4):320-334. doi: 10.1159/000520040. Epub 2021 Nov 26. J Innate Immun. 2022. PMID: 34839285 Free PMC article.
References
-
- Barclay J. W., Morgan A., Burgoyne R. D. (2005) Cell Calcium 38, 343–353 - PubMed
-
- Burgoyne R. D., Morgan A. (2003) Physiol. Rev. 83, 581–632 - PubMed
-
- Mayorga L. S., Tomes C. N., Belmonte S. A. (2007) IUBMB Life 59, 286–292 - PubMed
-
- Yanagimachi R. (1994) in The Physiology of Reproduction (Knobil E., Neill J. D. eds) pp. 189–317, Raven Press, New York
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

