The effect on intracytoplasmic sperm injection outcome of genotype, male germ cell stage and freeze-thawing in mice
- PMID: 20552034
- PMCID: PMC2884038
- DOI: 10.1371/journal.pone.0011062
The effect on intracytoplasmic sperm injection outcome of genotype, male germ cell stage and freeze-thawing in mice
Abstract
Background: Intracytoplasmic sperm injection (ICSI) has been widely used to study the mechanisms of mammalian fertilization and to rescue male-factor infertility in humans and animals. However, very few systematic analyses have been conducted to define factors affecting the efficiency of ICSI. In this study, we undertook a large-scale series of ICSI experiments in mice to define the factors that might affect outcomes.
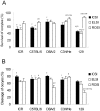
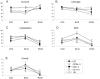
Methodology/principal findings: We used a 5 x 3 x 2 factorial design with the following factors: mouse genotype (ICR, C57BL/6, DBA/2, C3H/He, and 129/Sv strains), type of male germ cells (epididymal sperm, elongated or round spermatids), and their freeze-thawing treatment. The efficiencies (parameters) of each developmental step were analyzed by three-way ANOVA (significance level P<0.01). The type of male germ cells affected all the four parameters observed: oocyte survival after injection, cleavage of oocytes, implantation, and birth of offspring. Genotype affected the oocyte survival, cleavage and birth rates, whereas freeze-thawing had no effects on any of the parameters. There were significant genotype/cell type interactions for oocyte survival and cleavage, indicating that they were determined by a combination of strain and germ cell maturity. Multiple comparisons revealed that spermatozoa and elongated spermatids gave better implantation and birth rates than did round spermatids, while spermatozoa and elongated spermatozoa were indistinguishable in their ability to support embryonic development. The best overall efficiency (birth rate per oocytes injected) was obtained with frozen-thawed DBA/2 strain elongated spermatids (23.2+/-4.2%).
Conclusions/significance: The present study provides the first comprehensive information on ICSI using the mouse as a model and will contribute to the efficient use of materials, time, and efforts in biomedical research and clinics involving ICSI.
Conflict of interest statement
Figures
Similar articles
-
Development of feline embryos produced by Piezo-actuated intracytoplasmic sperm injection of elongated spermatids.J Reprod Dev. 2019 Jun 14;65(3):245-250. doi: 10.1262/jrd.2018-119. Epub 2019 Feb 18. J Reprod Dev. 2019. PMID: 30773507 Free PMC article.
-
Similar time restriction for intracytoplasmic sperm injection and round spermatid injection into activated oocytes for efficient offspring production.Biol Reprod. 2004 Jun;70(6):1863-9. doi: 10.1095/biolreprod.103.025171. Epub 2004 Feb 25. Biol Reprod. 2004. PMID: 14985245
-
Comparison of pre- and post-implantation development following the application of three artificial activating stimuli in a mouse model with round-headed sperm cells deficient for oocyte activation.Hum Reprod. 2013 May;28(5):1190-8. doi: 10.1093/humrep/det038. Epub 2013 Mar 12. Hum Reprod. 2013. PMID: 23482335
-
Intracytoplasmic sperm injection experiments using the mouse as a model.Hum Reprod. 1998 Apr;13 Suppl 1:87-98. doi: 10.1093/humrep/13.suppl_1.87. Hum Reprod. 1998. PMID: 9663773 Review.
-
Fertilization and developmental initiation of oocytes by injection of spermatozoa and pre-spermatozoal cells.Ital J Anat Embryol. 2005;110(2 Suppl 1):145-50. Ital J Anat Embryol. 2005. PMID: 16101032 Review.
Cited by
-
Development of feline embryos produced by Piezo-actuated intracytoplasmic sperm injection of elongated spermatids.J Reprod Dev. 2019 Jun 14;65(3):245-250. doi: 10.1262/jrd.2018-119. Epub 2019 Feb 18. J Reprod Dev. 2019. PMID: 30773507 Free PMC article.
-
Progress and future prospect of in vitro spermatogenesis.Oncotarget. 2017 Jul 27;8(39):66709-66727. doi: 10.18632/oncotarget.19640. eCollection 2017 Sep 12. Oncotarget. 2017. PMID: 29029549 Free PMC article. Review.
-
Viable offspring obtained from Prm1-deficient sperm in mice.Sci Rep. 2016 Jun 2;6:27409. doi: 10.1038/srep27409. Sci Rep. 2016. PMID: 27250771 Free PMC article.
-
Improved development of mouse somatic cell nuclear transfer embryos by chlamydocin analogues, class I and IIa histone deacetylase inhibitors†.Biol Reprod. 2021 Aug 3;105(2):543-553. doi: 10.1093/biolre/ioab096. Biol Reprod. 2021. PMID: 33982061 Free PMC article.
-
EPC1/TIP60-Mediated Histone Acetylation Facilitates Spermiogenesis in Mice.Mol Cell Biol. 2017 Sep 12;37(19):e00082-17. doi: 10.1128/MCB.00082-17. Print 2017 Oct 1. Mol Cell Biol. 2017. PMID: 28694333 Free PMC article.
References
-
- Uehara T, Yanagimachi R. Microsurgical injection of spermatozoa into hamster eggs with subsequent transformation of sperm nuclei into male pronuclei. Biol Reprod. 1976;15:467–470. - PubMed
-
- Uehara T, Yanagimachi R. Behavior of nuclei of testicular, caput and cauda epididymal spermatozoa injected into hamster eggs. Biol Reprod. 1977;16:315–321. - PubMed
-
- Yanagimachi R. Intracytoplasmic injection of spermatozoa and spermatogenic cells: its biology and applications in humans and animals. Reprod Biomed Online. 2005;10:247–288. - PubMed
-
- Ogura A, Ogonuki N, Miki H, Inoue K. Microinsemination and nuclear transfer using male germ cells. Int Rev Cytol. 2005;246:189–229. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

