A thalamo-hippocampal-ventral tegmental area loop may produce the positive feedback that underlies the psychotic break in schizophrenia
- PMID: 20553749
- PMCID: PMC3507433
- DOI: 10.1016/j.biopsych.2010.04.007
A thalamo-hippocampal-ventral tegmental area loop may produce the positive feedback that underlies the psychotic break in schizophrenia
Abstract
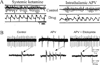
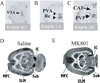
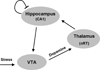
The N-methyl-D-aspartate receptor (NMDAR) hypofunction model of schizophrenia is based on the ability of NMDAR antagonists to produce many symptoms of the disease. Recent work in rats shows that NMDAR antagonist works synergistically with dopamine to produce delta frequency bursting in the thalamus. This finding, together with other results in the literature, suggests a mechanism for the sudden onset of schizophrenia. Among the thalamic nuclei most activated by NMDAR antagonist is the nucleus reuniens. This nucleus excites the cornu ammonis area 1 (CA1) region of the hippocampus. Experiments indicate that such activation can lead to excitation of dopaminergic cells of the ventral tegmental area by a polysynaptic pathway. The resulting elevation of dopamine in the thalamus will enhance thalamic bursting, thereby creating a loop with the potential for positive feedback. We show through computer simulations that in individuals with susceptibility to schizophrenia (e.g., because of partially compromised NMDAR function), an event that stimulates the dopamine system, such as stress, can cause the system to reach the threshold for thalamic bursting. When this occurs, positive feedback in the loop will cause all components to become highly active and to remain active after the triggering stimulus is removed. This is a physiologically specific hypothesis for the sudden and lasting transition that underlies the psychotic break in schizophrenia. Furthermore, the model provides an explanation for the observed selective activation of the CA1 hippocampal region in schizophrenia. The model also predicts an increase of basal activity in the dopamine system and thalamus; the relevant evidence is reviewed.
Copyright 2010 Society of Biological Psychiatry. Published by Elsevier Inc. All rights reserved.
Figures

Comment in
-
Dopamine in the thalamus: a hotbed for psychosis?Biol Psychiatry. 2010 Jul 1;68(1):3-4. doi: 10.1016/j.biopsych.2010.05.014. Biol Psychiatry. 2010. PMID: 20609835 Free PMC article. No abstract available.
Similar articles
-
NMDAR antagonist action in thalamus imposes δ oscillations on the hippocampus.J Neurophysiol. 2012 Jun;107(11):3181-9. doi: 10.1152/jn.00072.2012. Epub 2012 Mar 14. J Neurophysiol. 2012. PMID: 22423006 Free PMC article.
-
The role of dopamine-dependent negative feedback in the hippocampus-basal ganglia-thalamus-hippocampus loop in the extinction of responses.Neurosci Behav Physiol. 2008 May;38(4):399-405. doi: 10.1007/s11055-008-0057-4. Neurosci Behav Physiol. 2008. PMID: 18401733 Review.
-
Convergent Inputs from the Hippocampus and Thalamus to the Nucleus Accumbens Regulate Dopamine Neuron Activity.J Neurosci. 2018 Dec 12;38(50):10607-10618. doi: 10.1523/JNEUROSCI.2629-16.2018. Epub 2018 Oct 24. J Neurosci. 2018. PMID: 30355626 Free PMC article.
-
Activation of dopaminergic neurotransmission in the medial prefrontal cortex by N-methyl-d-aspartate stimulation of the ventral hippocampus in rats.Neuroscience. 2005;132(1):219-32. doi: 10.1016/j.neuroscience.2004.12.016. Neuroscience. 2005. PMID: 15780480
-
NMDA receptor hypofunction and the thalamus in schizophrenia.Physiol Behav. 2014 May 28;131:156-9. doi: 10.1016/j.physbeh.2014.04.038. Epub 2014 May 2. Physiol Behav. 2014. PMID: 24792662 Review.
Cited by
-
Abnormal stress responsivity in a rodent developmental disruption model of schizophrenia.Neuropsychopharmacology. 2013 Oct;38(11):2131-9. doi: 10.1038/npp.2013.110. Epub 2013 May 8. Neuropsychopharmacology. 2013. PMID: 23652286 Free PMC article.
-
Correlation Between Levels of Delusional Beliefs and Perfusion of the Hippocampus and an Associated Network in a Non-Help-Seeking Population.Biol Psychiatry Cogn Neurosci Neuroimaging. 2018 Feb;3(2):178-186. doi: 10.1016/j.bpsc.2017.06.007. Epub 2017 Jul 13. Biol Psychiatry Cogn Neurosci Neuroimaging. 2018. PMID: 29529413 Free PMC article.
-
Prefrontal-Thalamic Anatomical Connectivity and Executive Cognitive Function in Schizophrenia.Biol Psychiatry. 2018 Mar 15;83(6):509-517. doi: 10.1016/j.biopsych.2017.09.022. Epub 2017 Sep 28. Biol Psychiatry. 2018. PMID: 29113642 Free PMC article.
-
The Nucleus Reuniens of the Midline Thalamus Gates Prefrontal-Hippocampal Modulation of Ventral Tegmental Area Dopamine Neuron Activity.J Neurosci. 2016 Aug 24;36(34):8977-84. doi: 10.1523/JNEUROSCI.1402-16.2016. J Neurosci. 2016. PMID: 27559178 Free PMC article.
-
Schizophrenia Exhibits Bi-directional Brain-Wide Alterations in Cortico-Striato-Cerebellar Circuits.Cereb Cortex. 2019 Dec 17;29(11):4463-4487. doi: 10.1093/cercor/bhy306. Cereb Cortex. 2019. PMID: 31157363 Free PMC article.
References
-
- Olney JW, Newcomer JW, Farber NB. NMDA receptor hypofunction model of schizophrenia. J Psychiatr Res. 1999;33(6):523–533. - PubMed
-
- Umbricht D, et al. Ketamine-induced deficits in auditory and visual context-dependent processing in healthy volunteers: implications for models of cognitive deficits in schizophrenia. Arch Gen Psychiatry. 2000;57(12):1139–1147. - PubMed
-
- Abi-Saab WM, D'Souza DC, Moghaddam B, Krystal JH. The NMDA antagonist model for schizophrenia: promise and pitfalls. Pharmacopsychiatry. 1998;31(Suppl 2):104–109. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

