Plakophilin 2 couples actomyosin remodeling to desmosomal plaque assembly via RhoA
- PMID: 20554761
- PMCID: PMC2921118
- DOI: 10.1091/mbc.E10-02-0131
Plakophilin 2 couples actomyosin remodeling to desmosomal plaque assembly via RhoA
Erratum in
- Mol Biol Cell. 2010 Oct 15;21(20):3615
Abstract
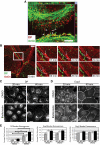
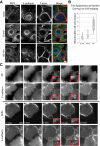
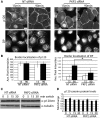
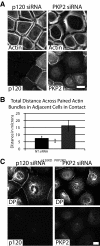
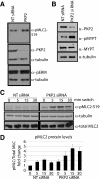
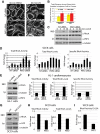
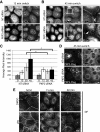
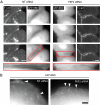
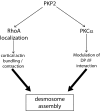
Plakophilin 2 (PKP2), an armadillo family member closely related to p120 catenin (p120ctn), is a constituent of the intercellular adhesive junction, the desmosome. We previously showed that PKP2 loss prevents the incorporation of desmosome precursors enriched in the plaque protein desmoplakin (DP) into newly forming desmosomes, in part by disrupting PKC-dependent regulation of DP assembly competence. On the basis of the observation that DP incorporation into junctions is cytochalasin D-sensitive, here we ask whether PKP2 may also contribute to actin-dependent regulation of desmosome assembly. We demonstrate that PKP2 knockdown impairs cortical actin remodeling after cadherin ligation, without affecting p120ctn expression or localization. Our data suggest that these defects result from the failure of activated RhoA to localize at intercellular interfaces after cell-cell contact and an elevation of cellular RhoA, stress fibers, and other indicators of contractile signaling in squamous cell lines and atrial cardiomyocytes. Consistent with these observations, RhoA activation accelerated DP redistribution to desmosomes during the first hour of junction assembly, whereas sustained RhoA activity compromised desmosome plaque maturation. Together with our previous findings, these data suggest that PKP2 may functionally link RhoA- and PKC-dependent pathways to drive actin reorganization and regulate DP-IF interactions required for normal desmosome assembly.
Figures

Similar articles
-
Plakophilin 3 mediates Rap1-dependent desmosome assembly and adherens junction maturation.Mol Biol Cell. 2014 Nov 15;25(23):3749-64. doi: 10.1091/mbc.E14-05-0968. Epub 2014 Sep 10. Mol Biol Cell. 2014. PMID: 25208567 Free PMC article.
-
Plakophilin 2: a critical scaffold for PKC alpha that regulates intercellular junction assembly.J Cell Biol. 2008 May 19;181(4):605-13. doi: 10.1083/jcb.200712133. Epub 2008 May 12. J Cell Biol. 2008. PMID: 18474624 Free PMC article.
-
Palmitoylation of plakophilin is required for desmosome assembly.J Cell Sci. 2014 Sep 1;127(Pt 17):3782-93. doi: 10.1242/jcs.149849. Epub 2014 Jul 7. J Cell Sci. 2014. PMID: 25002405 Free PMC article.
-
Plakophilins: Multifunctional proteins or just regulators of desmosomal adhesion?Biochim Biophys Acta. 2007 Jan;1773(1):69-77. doi: 10.1016/j.bbamcr.2006.04.009. Epub 2006 May 6. Biochim Biophys Acta. 2007. PMID: 16765467 Review.
-
Plakophilins in desmosomal adhesion and signaling.Cell Commun Adhes. 2014 Feb;21(1):25-42. doi: 10.3109/15419061.2013.876017. Cell Commun Adhes. 2014. PMID: 24460199 Review.
Cited by
-
Dsg2 ectodomain organization increases throughout desmosome assembly.Cell Adh Migr. 2024 Dec;18(1):1-13. doi: 10.1080/19336918.2024.2333366. Epub 2024 Apr 2. Cell Adh Migr. 2024. PMID: 38566311 Free PMC article.
-
SIRT1 activation and its effect on intercalated disc proteins as a way to reduce doxorubicin cardiotoxicity.Front Pharmacol. 2022 Nov 4;13:1035387. doi: 10.3389/fphar.2022.1035387. eCollection 2022. Front Pharmacol. 2022. PMID: 36408244 Free PMC article. Review.
-
The keratin-desmosome scaffold: pivotal role of desmosomes for keratin network morphogenesis.Cell Mol Life Sci. 2020 Feb;77(3):543-558. doi: 10.1007/s00018-019-03198-y. Epub 2019 Jun 26. Cell Mol Life Sci. 2020. PMID: 31243490 Free PMC article.
-
Autoantibody-Specific Signalling in Pemphigus.Front Med (Lausanne). 2021 Aug 9;8:701809. doi: 10.3389/fmed.2021.701809. eCollection 2021. Front Med (Lausanne). 2021. PMID: 34434944 Free PMC article. Review.
-
The impact of signaling pathways on the desmosome ultrastructure in pemphigus.Front Immunol. 2025 Jan 15;15:1497241. doi: 10.3389/fimmu.2024.1497241. eCollection 2024. Front Immunol. 2025. PMID: 39882246 Free PMC article.
References
-
- Anastasiadis P. Z., Moon S. Y., Thoreson M. A., Mariner D. J., Crawford H. C., Zheng Y., Reynolds A. B. Inhibition of RhoA by p120 catenin. Nat. Cell Biol. 2000;2:637–644. - PubMed
-
- Anastasiadis P. Z., Reynolds A. B. Regulation of Rho GTPases by p120-catenin. Curr. Opin. Cell Biol. 2001;13:604–610. - PubMed
-
- Angst B. D., Nilles L. A., Green K. J. Desmoplakin II expression is not restricted to stratified epithelia. J. Cell Sci. 1990;97(Pt 2):247–257. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

