Multiple endocrine neoplasia type 1 deletion in pancreatic alpha-cells leads to development of insulinomas in mice
- PMID: 20555035
- PMCID: PMC2940531
- DOI: 10.1210/en.2009-1251
Multiple endocrine neoplasia type 1 deletion in pancreatic alpha-cells leads to development of insulinomas in mice
Abstract
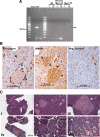
The pancreatic alpha- and beta-cells are critical components in regulating blood glucose homeostasis via secretion of glucagon and insulin, respectively. Both cell types are typically localized in the islets of Langerhans. However, little is known about the roles of paracrine interactions that contribute to their physiological functions. The lack of suitable cell lines to study alpha- and beta-cells interactions have led us to develop an alpha-cell-specific Cre-expressing transgenic line utilizing a glucagon promoter sequence, the Glu-Cre transgenic mouse. Here, we demonstrate that the Glu-Cre could specifically and efficiently excise floxed target genes in adult islet alpha-cells. We further showed that deletion of the tumor suppressor gene, multiple endocrine neoplasia type 1 (Men1), in alpha-cells led to tumorigenesis. However, to our surprise, the lack of Men1 in alpha-cells did not result in glucagonomas but rather beta-cell insulinomas. Because deletion of the Men1 alleles was only present in alpha-cells, our data suggested that cross communication between alpha- and beta-cells contributes to tumorigenesis in the absence of Men1. Together, we believed that the new model systems described here will allow future studies to decipher cellular interactions between islet alpha- and beta-cells in a physiological context.
Figures



References
-
- Gromada J, Franklin I, Wollheim CB 2007 α-Cells of the endocrine pancreas: 35 years of research but the enigma remains. Endocr Rev 28:84–116 - PubMed
-
- Bonner-Weir S, Orci L 1982 New perspectives on the microvasculature of the islets of Langerhans in the rat. Diabetes 31:883–889 - PubMed
-
- Chandrasekharappa SC, Guru SC, Manickam P, Olufemi SE, Collins FS, Emmert-Buck MR, Debelenko LV, Zhuang Z, Lubensky IA, Liotta LA, Crabtree JS, Wang Y, Roe BA, Weisemann J, Boguski MS, Agarwal SK, Kester MB, Kim YS, Heppner C, Dong Q, Spiegel AM, Burns AL, Marx SJ 1997 Positional cloning of the gene for multiple endocrine neoplasia-type 1. Science 276:404–407 - PubMed
-
- Lemmens I, Van de Ven WJ, Kas K, Zhang CX, Giraud S, Wautot V, Buisson N, De Witte K, Salandre J, Lenoir G, Pugeat M, Calender A, Parente F, Quincey D, Gaudray P, De Wit MJ, Lips CJ, Höppener JW, Khodaei S, Grant AL, Weber G, Kytölä S, Teh BT, Farnebo F, Thakker RV 1997 Identification of the multiple endocrine neoplasia type 1 (MEN1) gene. The European consortium on MEN1. Hum Mol Genet 6:1177–1183 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

