The multifunctional PE_PGRS11 protein from Mycobacterium tuberculosis plays a role in regulating resistance to oxidative stress
- PMID: 20558725
- PMCID: PMC2945531
- DOI: 10.1074/jbc.M110.135251
The multifunctional PE_PGRS11 protein from Mycobacterium tuberculosis plays a role in regulating resistance to oxidative stress
Erratum in
-
Correction: The multifunctional PE_PGRS11 protein from Mycobacterium tuberculosis plays a role in regulating resistance to oxidative stress.J Biol Chem. 2019 Dec 13;294(50):19445. doi: 10.1074/jbc.AAC119.011906. J Biol Chem. 2019. PMID: 31836672 Free PMC article. No abstract available.
Abstract
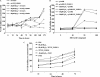
Mycobacterium tuberculosis utilizes unique strategies to survive amid the hostile environment of infected host cells. Infection-specific expression of a unique mycobacterial cell surface antigen that could modulate key signaling cascades can act as a key survival strategy in curtailing host effector responses like oxidative stress. We demonstrate here that hypothetical PE_PGRS11 ORF encodes a functional phosphoglycerate mutase. The transcriptional analysis revealed that PE_PGRS11 is a hypoxia-responsive gene, and enforced expression of PE_PGRS11 by recombinant adenovirus or Mycobacterium smegmatis imparted resistance to alveolar epithelial cells against oxidative stress. PE_PGRS11-induced resistance to oxidative stress necessitated the modulation of genetic signatures like induced expression of Bcl2 or COX-2. This modulation of specific antiapoptotic molecular signatures involved recognition of PE_PGRS11 by TLR2 and subsequent activation of the PI3K-ERK1/2-NF-κB signaling axis. Furthermore, PE_PGRS11 markedly diminished H(2)O(2)-induced p38 MAPK activation. Interestingly, PE_PGRS11 protein was exposed at the mycobacterial cell surface and was involved in survival of mycobacteria under oxidative stress. Furthermore, PE_PGRS11 displayed differential B cell responses during tuberculosis infection. Taken together, our investigation identified PE_PGRS11 as an in vivo expressed immunodominant antigen that plays a crucial role in modulating cellular life span restrictions imposed during oxidative stress by triggering TLR2-dependent expression of COX-2 and Bcl2. These observations clearly provide a mechanistic basis for the rescue of pathogenic Mycobacterium-infected lung epithelial cells from oxidative stress.
Figures


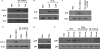

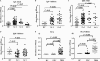
Similar articles
-
ESAT-6 induced COX-2 expression involves coordinated interplay between PI3K and MAPK signaling.Mol Immunol. 2012 Jan;49(4):655-63. doi: 10.1016/j.molimm.2011.11.011. Epub 2011 Dec 10. Mol Immunol. 2012. PMID: 22154837
-
Execution of macrophage apoptosis by PE_PGRS33 of Mycobacterium tuberculosis is mediated by Toll-like receptor 2-dependent release of tumor necrosis factor-alpha.J Biol Chem. 2007 Jan 12;282(2):1039-50. doi: 10.1074/jbc.M604379200. Epub 2006 Nov 9. J Biol Chem. 2007. PMID: 17095513
-
Src homology 3-interacting domain of Rv1917c of Mycobacterium tuberculosis induces selective maturation of human dendritic cells by regulating PI3K-MAPK-NF-kappaB signaling and drives Th2 immune responses.J Biol Chem. 2010 Nov 19;285(47):36511-22. doi: 10.1074/jbc.M110.158055. Epub 2010 Sep 13. J Biol Chem. 2010. PMID: 20837474 Free PMC article.
-
PE_PGRS: Vital proteins in promoting mycobacterial survival and modulating host immunity and metabolism.Cell Microbiol. 2021 Mar;23(3):e13290. doi: 10.1111/cmi.13290. Epub 2020 Dec 1. Cell Microbiol. 2021. PMID: 33217152 Review.
-
PE_PGRS33, an Important Virulence Factor of Mycobacterium tuberculosis and Potential Target of Host Humoral Immune Response.Cells. 2021 Jan 15;10(1):161. doi: 10.3390/cells10010161. Cells. 2021. PMID: 33467487 Free PMC article. Review.
Cited by
-
Inferring gene function from evolutionary change in signatures of translation efficiency.Genome Biol. 2014 Mar 3;15(3):R44. doi: 10.1186/gb-2014-15-3-r44. Genome Biol. 2014. PMID: 24580753 Free PMC article.
-
Delineating the Physiological Roles of the PE and Catalytic Domains of LipY in Lipid Consumption in Mycobacterium-Infected Foamy Macrophages.Infect Immun. 2018 Aug 22;86(9):e00394-18. doi: 10.1128/IAI.00394-18. Print 2018 Sep. Infect Immun. 2018. PMID: 29986895 Free PMC article.
-
Differential cellular recognition pattern to M. tuberculosis targets defined by IFN-γ and IL-17 production in blood from TB + patients from Honduras as compared to health care workers: TB and immune responses in patients from Honduras.BMC Infect Dis. 2013 Mar 6;13:125. doi: 10.1186/1471-2334-13-125. BMC Infect Dis. 2013. PMID: 23497342 Free PMC article.
-
Role of C-terminal domain of Mycobacterium tuberculosis PE6 (Rv0335c) protein in host mitochondrial stress and macrophage apoptosis.Apoptosis. 2023 Feb;28(1-2):136-165. doi: 10.1007/s10495-022-01778-1. Epub 2022 Oct 18. Apoptosis. 2023. PMID: 36258102 Free PMC article.
-
PPE38 modulates the innate immune response and is required for Mycobacterium marinum virulence.Infect Immun. 2012 Jan;80(1):43-54. doi: 10.1128/IAI.05249-11. Epub 2011 Oct 28. Infect Immun. 2012. PMID: 22038915 Free PMC article.
References
-
- Józefowski S., Sobota A., Kwiatkowska K. (2008) BioEssays 30, 943–954 - PubMed
-
- Andersen P., Doherty T. M. (2005) Nat. Rev. Microbiol. 3, 656–662 - PubMed
-
- Adindla S., Guruprasad L. (2003) J. Biosci. 28, 169–179 - PubMed
-
- Cole S. T., Brosch R., Parkhill J., Garnier T., Churcher C., Harris D., Gordon S. V., Eiglmeier K., Gas S., Barry C. E., 3rd, Tekaia F., Badcock K., Basham D., Brown D., Chillingworth T., Connor R., Davies R., Devlin K., Feltwell T., Gentles S., Hamlin N., Holroyd S., Hornsby T., Jagels K., Krogh A., McLean J., Moule S., Murphy L., Oliver K., Osborne J., Quail M. A., Rajandream M. A., Rogers J., Rutter S., Seeger K., Skelton J., Squares R., Squares S., Sulston J. E., Taylor K., Whitehead S., Barrell B. G. (1998) Nature 393, 537–544 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

