Thioredoxin-interacting protein (Txnip) gene expression: sensing oxidative phosphorylation status and glycolytic rate
- PMID: 20558747
- PMCID: PMC2919144
- DOI: 10.1074/jbc.M110.108290
Thioredoxin-interacting protein (Txnip) gene expression: sensing oxidative phosphorylation status and glycolytic rate
Abstract
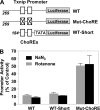
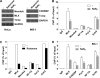
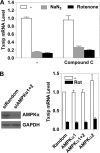
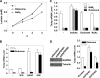
Thioredoxin-interacting protein (Txnip) has important functions in regulating cellular metabolism including glucose utilization; the expression of the Txnip gene is sensitive to the availability of glucose and other fuels. Here, we show that Txnip expression is down-regulated at the transcriptional level by diverse inhibitors of mitochondrial oxidative phosphorylation (OXPHOS). The effect of these OXPHOS inhibitors is mediated by earlier identified carbohydrate-response elements (ChoREs) on the Txnip promoter and the ChoRE-associated transcription factors Max-like protein X (MLX) and MondoA (or carbohydrate-response element-binding protein (ChREBP)) involved in glucose-induced Txnip expression, suggesting that inhibited oxidative phosphorylation compromises glucose-induced effects on Txnip expression. We also show that the OXPHOS inhibitors repress the Txnip transcription most likely by inducing the glycolytic rate, and increased glycolytic flux decreases the levels of glycolytic intermediates important for the function of MLX and MondoA (or ChREBP). Our findings suggest that the Txnip expression is tightly correlated with glycolytic flux, which is regulated by oxidative phosphorylation status. The identified link between the Txnip expression and glycolytic activity implies a mechanism by which the cellular glucose uptake/homeostasis is regulated in response to various metabolic cues, oxidative phosphorylation status, and other physiological signals, and this may facilitate our efforts toward understanding metabolism in normal or cancer cells.
Figures




References
-
- Kim S. Y., Suh H. W., Chung J. W., Yoon S. R., Choi I. (2007) Cell Mol. Immunol. 4, 345–351 - PubMed
-
- Chen C. L., Lin C. F., Chang W. T., Huang W. C., Teng C. F., Lin Y. S. (2008) Blood 111, 4365–4374 - PubMed
-
- Han S. H., Jeon J. H., Ju H. R., Jung U., Kim K. Y., Yoo H. S., Lee Y. H., Song K. S., Hwang H. M., Na Y. S., Yang Y., Lee K. N., Choi I. (2003) Oncogene 22, 4035–4046 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

