Membrane blebbing as an assessment of functional rescue of dysferlin-deficient human myotubes via nonsense suppression
- PMID: 20558759
- PMCID: PMC3774516
- DOI: 10.1152/japplphysiol.01366.2009
Membrane blebbing as an assessment of functional rescue of dysferlin-deficient human myotubes via nonsense suppression
Abstract
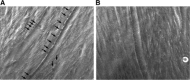
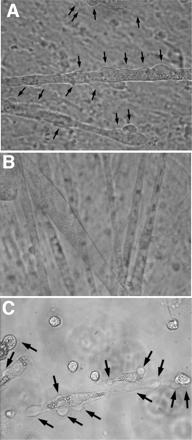
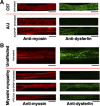
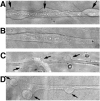
Mutations that result in the loss of the protein dysferlin result in defective muscle membrane repair and cause either a form of limb girdle muscular dystrophy (type 2B) or Miyoshi myopathy. Most patients are compound heterozygotes, often carrying one allele with a nonsense mutation. Using dysferlin-deficient mouse and human myocytes, we demonstrated that membrane blebbing in skeletal muscle myotubes in response to hypotonic shock requires dysferlin. Based on this, we developed an in vitro assay to assess rescue of dysferlin function in skeletal muscle myotubes. This blebbing assay may be useful for drug discovery/validation for dysferlin deficiency. With this assay, we demonstrate that the nonsense suppression drug, ataluren (PTC124), is able to induce read-through of the premature stop codon in a patient with a R1905X mutation in dysferlin and produce sufficient functional dysferlin (approximately 15% of normal levels) to rescue myotube membrane blebbing. Thus ataluren is a potential therapeutic for dysferlin-deficient patients harboring nonsense mutations.
Figures

References
-
- Bansal D, Campbell KP. Dysferlin and the plasma membrane repair in muscular dystrophy. Trends Cell Biol 14: 206–213, 2004 - PubMed
-
- Bansal D, Miyake K, Vogel SS, Groh S, Chen CC, Williamson R, McNeil PL, Campbell KP. Defective membrane repair in dysferlin-deficient muscular dystrophy. Nature 423: 168–172, 2003 - PubMed
-
- Bashir R, Britton S, Strachan T, Keers S, Vafiadaki E, Lako M, Richard I, Marchand S, Bourg N, Argov Z, Sadeh M, Mahjneh I, Marconi G, Passos-Bueno MR, Moreira Ede S, Zatz M, Beckmann JS, Bushby K. A gene related to Caenorhabditis elegans spermatogenesis factor fer-1 is mutated in limb-girdle muscular dystrophy type 2B. Nat Genet 20: 37–42, 1998 - PubMed
-
- Bittner RE, Anderson LV, Burkhardt E, Bashir R, Vafiadaki E, Ivanova S, Raffelsberger T, Maerk I, Höger H, Jung M, Karbasiyan M, Storch M, Lassmann H, Moss JA, Davison K, Harrison R, Bushby KM, Reis A. Dysferlin deletion in SJL mice (SJL-Dysf) defines a natural model for limb girdle muscular dystrophy 2B. Nat Genet 23: 141–142, 1999 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

