Endogenous Wnt/beta-catenin signaling is required for cardiac differentiation in human embryonic stem cells
- PMID: 20559569
- PMCID: PMC2886114
- DOI: 10.1371/journal.pone.0011134
Endogenous Wnt/beta-catenin signaling is required for cardiac differentiation in human embryonic stem cells
Abstract
Background: Wnt/beta-catenin signaling is an important regulator of differentiation and morphogenesis that can also control stem cell fates. Our group has developed an efficient protocol to generate cardiomyocytes from human embryonic stem (ES) cells via induction with activin A and BMP4.
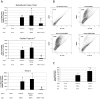
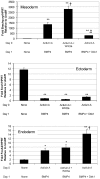
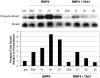
Methodology/principal findings: We tested the hypothesis that Wnt/beta-catenin signals control both early mesoderm induction and later cardiac differentiation in this system. Addition of exogenous Wnt3a at the time of induction enhanced cardiac differentiation, while early inhibition of endogenous Wnt/beta-catenin signaling with Dkk1 inhibited cardiac differentiation, as indicated by quantitative RT-PCR analysis for beta-myosin heavy chain (beta-MHC), cardiac troponin T (cTnT), Nkx2.5, and flow cytometry analysis for sarcomeric myosin heavy chain (sMHC). Conversely, late antagonism of endogenously produced Wnts enhanced cardiogenesis, indicating a biphasic role for the pathway in human cardiac differentiation. Using quantitative RT-PCR, we show that canonical Wnt ligand expression is induced by activin A/BMP4 treatment, and the extent of early Wnt ligand expression can predict the subsequent efficiency of cardiogenesis. Measurement of Brachyury expression showed that addition of Wnt3a enhances mesoderm induction, whereas blockade of endogenously produced Wnts markedly inhibits mesoderm formation. Finally, we show that Wnt/beta-catenin signaling is required for Smad1 activation by BMP4.
Conclusions/significance: Our data indicate that induction of mesoderm and subsequent cardiac differentiation from human ES cells requires fine-tuned cross talk between activin A/BMP4 and Wnt/beta-catenin pathways. Controlling these pathways permits efficient generation of cardiomyocytes for basic studies or cardiac repair applications.
Conflict of interest statement
Figures

Similar articles
-
Biphasic role for Wnt/beta-catenin signaling in cardiac specification in zebrafish and embryonic stem cells.Proc Natl Acad Sci U S A. 2007 Jun 5;104(23):9685-90. doi: 10.1073/pnas.0702859104. Epub 2007 May 23. Proc Natl Acad Sci U S A. 2007. PMID: 17522258 Free PMC article.
-
Temporal and contextual orchestration of cardiac fate by WNT-BMP synergy and threshold.J Cell Mol Med. 2010 Aug;14(8):2094-108. doi: 10.1111/j.1582-4934.2009.00774.x. Epub 2010 May 11. J Cell Mol Med. 2010. PMID: 19432819 Free PMC article.
-
Defining early lineage specification of human embryonic stem cells by the orchestrated balance of canonical Wnt/beta-catenin, Activin/Nodal and BMP signaling.Development. 2008 Sep;135(17):2969-79. doi: 10.1242/dev.021121. Epub 2008 Jul 30. Development. 2008. PMID: 18667462
-
The multiple phases and faces of wnt signaling during cardiac differentiation and development.Circ Res. 2010 Jul 23;107(2):186-99. doi: 10.1161/CIRCRESAHA.110.221531. Circ Res. 2010. PMID: 20651295 Review.
-
Signaling pathways controlling second heart field development.Circ Res. 2009 Apr 24;104(8):933-42. doi: 10.1161/CIRCRESAHA.109.194464. Circ Res. 2009. PMID: 19390062 Review.
Cited by
-
Molecular Signatures and Networks of Cardiomyocyte Differentiation in Humans and Mice.Mol Ther Nucleic Acids. 2020 Sep 4;21:696-711. doi: 10.1016/j.omtn.2020.07.011. Epub 2020 Jul 10. Mol Ther Nucleic Acids. 2020. PMID: 32769060 Free PMC article.
-
Differentiation of Cardiomyocytes from Human Pluripotent Stem Cells Using Monolayer Culture.Biomark Insights. 2015 May 27;10(Suppl 1):71-6. doi: 10.4137/BMI.S20050. eCollection 2015. Biomark Insights. 2015. PMID: 26052225 Free PMC article. Review.
-
Myocardial regeneration of the failing heart.Heart Fail Rev. 2013 Nov;18(6):815-33. doi: 10.1007/s10741-012-9348-5. Heart Fail Rev. 2013. PMID: 23001638 Review.
-
Directed cardiomyocyte differentiation from human pluripotent stem cells by modulating Wnt/β-catenin signaling under fully defined conditions.Nat Protoc. 2013 Jan;8(1):162-75. doi: 10.1038/nprot.2012.150. Epub 2012 Dec 20. Nat Protoc. 2013. PMID: 23257984 Free PMC article.
-
Activin-A and Bmp4 levels modulate cell type specification during CHIR-induced cardiomyogenesis.PLoS One. 2015 Feb 23;10(2):e0118670. doi: 10.1371/journal.pone.0118670. eCollection 2015. PLoS One. 2015. PMID: 25706534 Free PMC article.
References
-
- Menasche P. Skeletal myoblasts as a therapeutic agent. Prog Cardiovasc Dis. 2007;50:7–17. - PubMed
-
- Murry CE, Keller G. Differentiation of embryonic stem cells to clinically relevant populations: lessons from embryonic development. Cell. 2008;132:661–680. - PubMed
-
- Fukuda K, Yuasa S. Stem cells as a source of regenerative cardiomyocytes. Circ Res. 2006;98:1002–1013. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

