One lipid, multiple functions: how various pools of PI(4,5)P(2) are created in the plasma membrane
- PMID: 20559679
- PMCID: PMC11115911
- DOI: 10.1007/s00018-010-0432-5
One lipid, multiple functions: how various pools of PI(4,5)P(2) are created in the plasma membrane
Abstract
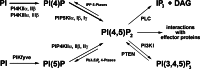
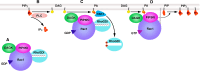
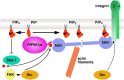
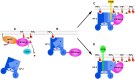
Phosphatidylinositol 4,5-bisphosphate [PI(4,5)P(2)] is a minor lipid of the inner leaflet of the plasma membrane that controls the activity of numerous proteins and serves as a source of second messengers. This multifunctionality of PI(4,5)P(2) relies on mechanisms ensuring transient appearance of PI(4,5)P(2) clusters in the plasma membrane. One such mechanism involves phosphorylation of PI(4)P to PI(4,5)P(2) by the type I phosphatidylinositol-4-phosphate 5-kinases (PIP5KI) at discrete membrane locations coupled with PI(4)P delivery/synthesis at the plasma membrane. Simultaneously, both PI(4)P and PI(4,5)P(2) participate in anchoring PIP5KI at the plasma membrane via electrostatic bonds. PIP5KI isoforms are also selectively recruited and activated at the plasma membrane by Rac1, talin, or AP-2 to generate PI(4,5)P(2) in ruffles and lamellipodia, focal contacts, and clathrin-coated pits. In addition, PI(4,5)P(2) can accumulate at sphingolipid/cholesterol-based rafts following activation of distinct membrane receptors or be sequestered in a reversible manner due to electrostatic constrains posed by proteins like MARCKS.
Figures

Similar articles
-
Loss of PIP5KIgamma, unlike other PIP5KI isoforms, impairs the integrity of the membrane cytoskeleton in murine megakaryocytes.J Clin Invest. 2008 Feb;118(2):812-9. doi: 10.1172/JCI34239. J Clin Invest. 2008. PMID: 18188447 Free PMC article.
-
Type I gamma phosphatidylinositol phosphate kinase targets and regulates focal adhesions.Nature. 2002 Nov 7;420(6911):89-93. doi: 10.1038/nature01082. Nature. 2002. PMID: 12422220
-
Phosphatidylinositol 4 phosphate regulates targeting of clathrin adaptor AP-1 complexes to the Golgi.Cell. 2003 Aug 8;114(3):299-310. doi: 10.1016/s0092-8674(03)00603-2. Cell. 2003. PMID: 12914695
-
Control of diverse subcellular processes by a single multi-functional lipid phosphatidylinositol 4,5-bisphosphate [PI(4,5)P2].Biochem J. 2016 Jun 15;473(12):1681-92. doi: 10.1042/BCJ20160069. Biochem J. 2016. PMID: 27288030 Free PMC article. Review.
-
Structure and Lateral Organization of Phosphatidylinositol 4,5-bisphosphate.Molecules. 2020 Aug 26;25(17):3885. doi: 10.3390/molecules25173885. Molecules. 2020. PMID: 32858905 Free PMC article. Review.
Cited by
-
PIP5Kγ Mediates PI(4,5)P2/Merlin/LATS1 Signaling Activation and Interplays with Hsc70 in Hippo-YAP Pathway Regulation.Int J Mol Sci. 2023 Sep 30;24(19):14786. doi: 10.3390/ijms241914786. Int J Mol Sci. 2023. PMID: 37834234 Free PMC article.
-
Structure and regulation of phospholipase Cβ and ε at the membrane.Chem Phys Lipids. 2021 Mar;235:105050. doi: 10.1016/j.chemphyslip.2021.105050. Epub 2021 Jan 7. Chem Phys Lipids. 2021. PMID: 33422547 Free PMC article. Review.
-
Phosphatidylinositol-5-phosphate 4-kinase gamma accumulates at the spindle pole and prevents microtubule depolymerization.Cell Div. 2019 Aug 21;14:9. doi: 10.1186/s13008-019-0053-9. eCollection 2019. Cell Div. 2019. PMID: 31452676 Free PMC article.
-
Phosphatidylinositol-4-phosphate 5-kinase and GEP100/Brag2 protein mediate antiangiogenic signaling by semaphorin 3E-plexin-D1 through Arf6 protein.J Biol Chem. 2011 Sep 30;286(39):34335-45. doi: 10.1074/jbc.M111.259499. Epub 2011 Jul 27. J Biol Chem. 2011. PMID: 21795701 Free PMC article.
-
Dual Regulation of R-Type CaV2.3 Channels by M1 Muscarinic Receptors.Mol Cells. 2016 Apr 30;39(4):322-9. doi: 10.14348/molcells.2016.2292. Epub 2016 Feb 26. Mol Cells. 2016. PMID: 26923189 Free PMC article.
References
-
- Hokin MR, Hokin LE. Enzyme secretion and the incorporation of P32 into phospholipides of pancreas slices. J Biol Chem. 1953;203:967–977. - PubMed
-
- Yin HL, Janmey PA. Phosphoinositide regulation of the actin cytoskeleton. Annu Rev Physiol. 2003;65:761–789. - PubMed
-
- Di Paolo SG, De Camilli P. Phosphoinositides in cell regulation and membrane dynamics. Nature. 2006;443:651–657. - PubMed
-
- Rameh LE, Cantley LC. The role of phosphoinositide 3-kinase lipid products in cell function. J Biol Chem. 1999;274:8347–8350. - PubMed
-
- Maehama T, Dixon JE. The tumor suppressor, PTEN/MMAC1, dephosphorylates the lipid second messenger, phosphatidylinositol 3,4,5-trisphosphate. J Biol Chem. 1998;273:13375–13378. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

