Type I IFN enhances follicular B cell contribution to the T cell-independent antibody response
- PMID: 20566717
- PMCID: PMC2901065
- DOI: 10.1084/jem.20092695
Type I IFN enhances follicular B cell contribution to the T cell-independent antibody response
Abstract
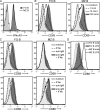
Humoral immunity to viruses and encapsulated bacteria is comprised of T cell-independent type 2 (TI-2) antibody responses that are characterized by rapid antibody production by marginal zone and B1 B cells. We demonstrate that toll-like receptor (TLR) ligands influence the TI-2 antibody response not only by enhancing the overall magnitude but also by skewing this response to one that is dominated by IgG isotypes. Importantly, TLR ligands facilitate this response by inducing type I interferon (IFN), which in turn elicits rapid and significant amounts of antigen-specific IgG2c predominantly from FO (follicular) B cells. Furthermore, we show that although the IgG2c antibody response requires B cell-autonomous IFN-alpha receptor signaling, it is independent of B cell-intrinsic TLR signaling. Thus, innate signals have the capacity to enhance TI-2 antibody responses by promoting participation of FO B cells, which then elaborate effective IgG anti-pathogen antibodies.
Figures
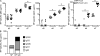




Similar articles
-
In vivo CD40-gp39 interactions are essential for thymus-dependent humoral immunity. I. In vivo expression of CD40 ligand, cytokines, and antibody production delineates sites of cognate T-B cell interactions.J Exp Med. 1993 Nov 1;178(5):1555-65. doi: 10.1084/jem.178.5.1555. J Exp Med. 1993. PMID: 7693849 Free PMC article.
-
Slc15a4 function is required for intact class switch recombination to IgG2c in response to TLR9 stimulation.Immunol Cell Biol. 2015 Feb;93(2):136-46. doi: 10.1038/icb.2014.82. Epub 2014 Oct 14. Immunol Cell Biol. 2015. PMID: 25310967
-
TLR signaling fine-tunes anti-influenza B cell responses without regulating effector T cell responses.J Immunol. 2007 Feb 15;178(4):2182-91. doi: 10.4049/jimmunol.178.4.2182. J Immunol. 2007. PMID: 17277123
-
Toll-like receptors and B-cell receptors synergize to induce immunoglobulin class-switch DNA recombination: relevance to microbial antibody responses.Crit Rev Immunol. 2010;30(1):1-29. doi: 10.1615/critrevimmunol.v30.i1.10. Crit Rev Immunol. 2010. PMID: 20370617 Free PMC article. Review.
-
B cell TLRs and induction of immunoglobulin class-switch DNA recombination.Front Biosci (Landmark Ed). 2012 Jun 1;17(7):2594-615. doi: 10.2741/4073. Front Biosci (Landmark Ed). 2012. PMID: 22652800 Free PMC article. Review.
Cited by
-
Immunogenicity and protective capacity of a virosomal respiratory syncytial virus vaccine adjuvanted with monophosphoryl lipid A in mice.PLoS One. 2012;7(5):e36812. doi: 10.1371/journal.pone.0036812. Epub 2012 May 9. PLoS One. 2012. PMID: 22590614 Free PMC article.
-
T-independent B-cell effect of agents associated with swine grower-finisher diarrhea.Vet Res Commun. 2024 Apr;48(2):991-1001. doi: 10.1007/s11259-023-10257-0. Epub 2023 Dec 4. Vet Res Commun. 2024. PMID: 38044397
-
Murine B cell development and antibody responses to model antigens are not impaired in the absence of the TNF receptor GITR.PLoS One. 2012;7(2):e31632. doi: 10.1371/journal.pone.0031632. Epub 2012 Feb 6. PLoS One. 2012. PMID: 22328941 Free PMC article.
-
An Adjuvant That Increases Protective Antibody Responses to Polysaccharide Antigens and Enables Recall Responses.J Infect Dis. 2019 Jan 7;219(2):323-334. doi: 10.1093/infdis/jiy506. J Infect Dis. 2019. PMID: 30289460 Free PMC article.
-
Single-cell RNA reveals a tumorigenic microenvironment in the interface zone of human breast tumors.Breast Cancer Res. 2023 Aug 29;25(1):100. doi: 10.1186/s13058-023-01703-7. Breast Cancer Res. 2023. PMID: 37644609 Free PMC article.
References
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

